What Metal Is In Catalytic Converters? Why Some Are Worth More
What Metal Is in Catalytic Converters?
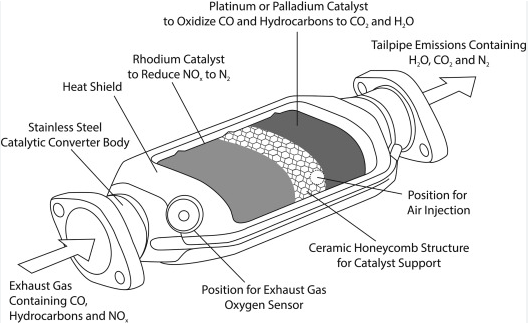
If you want the plain-English answer, catalytic converters mainly contain platinum, palladium, and rhodium. Those are the precious metals most people mean when they ask what metal is in catalytic converters. They are not packed inside as large solid pieces. In most designs, they are applied as a very thin catalytic coating on an internal honeycomb support, a structure described by Johnson Matthey.
Most catalytic converters get their value and function from thin coatings of platinum, palladium, and rhodium.
The Direct Answer in Plain English
So, what is in a catalytic converter? In simple terms, it is an exhaust-cleaning device that helps change harmful engine gases into less harmful emissions before they leave the tailpipe. If you are wondering what is a catalytic converter made of, the short answer is an outer metal shell plus an internal catalyst system, with platinum, palladium, and rhodium doing the chemical work.
Why These Metals Matter for Emissions Control
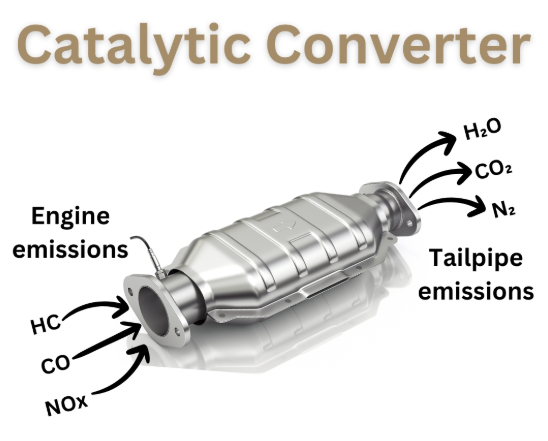
What do catalytic converters do in everyday driving? They help reduce pollutants such as carbon monoxide, unburned hydrocarbons, and nitrogen oxides. Plain-language explanations from HowStuffWorks show that these metals speed up exhaust reactions without being quickly used up. That is why what is the metal in catalytic converters matters not only to recyclers, but also to drivers, technicians, and anyone trying to understand repair costs or emissions systems.
- Platinum, palladium, and rhodium are the main active metals.
- They work as a coating, not as visible chunks you can pull out.
- Their job is to help convert harmful exhaust gases into less harmful ones.
- The exact metal recipe can change from one vehicle to another.
Why Not Every Converter Has the Same Metal Mix
Here is the part many people miss: what's in a catalytic converter is not always the same formula. Gasoline and diesel systems can use different catalyst strategies, and automakers also adjust the balance based on engine type, emissions rules, and durability goals. So when someone asks what is the metal in catalytic converters, the best answer is usually the same three names, but not always in the same proportion. And when people ask what's in a catalytic converter, they are usually talking about those active catalyst metals, not the rest of the hardware hidden inside.
What Is Inside a Catalytic Converter
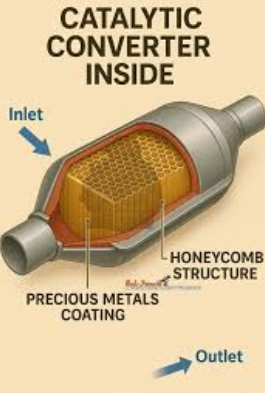
Those three precious metals do not sit inside the unit like loose chunks of metal. If you are wondering what is inside a catalytic converter, think of a layered exhaust device built to protect a very thin active surface. DieselNet describes converters as catalyst substrates coated with an active washcoat and packaged in steel housings. Johnson Matthey adds that the catalyst metals are typically nanoparticles coated on a substrate, sometimes called a brick.
The Shell, Substrate, and Washcoat
So, what's inside of a catalytic converter in plain English? First comes the outer shell, usually steel or stainless steel, which protects the converter from heat, vibration, moisture, and road conditions. Inside that housing sits the substrate, usually a ceramic or metallic honeycomb. Its job is to create a huge surface area while still letting exhaust flow through. On top of that substrate is the washcoat, a thin layer that helps hold and spread the active catalyst materials.
- Steel housing: protects the assembly and keeps the core in place.
- Mounting mat or insulation: cushions the core and helps manage heat.
- Ceramic or metallic substrate: forms honeycomb channels for exhaust flow.
- Washcoat: provides the surface that carries the active catalyst.
- Precious metals: platinum, palladium, and rhodium do the emissions-cleaning work.
Where the Precious Metals Actually Sit
When people ask what is the metal inside a catalytic converter, they usually mean the catalyst metals, not every material in the assembly. The important detail is where those metals sit. They are dispersed across the washcoat on the walls of the honeycomb channels, not stored as large visible pieces. That design gives exhaust gas far more contact with the active surface.
Why the Inside Design Affects Performance
The catalytic converter inside layout shapes how well the unit works. DieselNet highlights flow distribution, pressure drop, heat loss, and durability as key design concerns. In simple terms, the internal structure affects warm-up speed, how evenly gases reach the catalyst, and how well the converter survives real-world driving. That is why two units can look similar from the outside, yet differ a lot once you examine the inside of a catalytic converter and the way its metals are applied.
Platinum, Palladium, and Rhodium Side by Side
The names are familiar, but they do not do the same job. If someone asks what precious metal is in a catalytic converter, the honest answer is usually three metals working together, not one star ingredient. There is no single precious metal in catalytic converter designs. Platinum, palladium, and rhodium sit on the converter's internal coating, and each one helps clean a different part of the exhaust stream. In simple terms, platinum and palladium mostly support oxidation reactions, while rhodium is best known for reduction.
Platinum and Its Oxidation Role
Platinum helps convert carbon monoxide and unburned hydrocarbons into less harmful emissions. Phoenix Refining describes platinum as active in both gasoline and diesel converters, while Global Ardour notes its versatility makes it especially valuable in many diesel applications. Many searches for catalytic converter platinum, or even the shorthand cat converter platinum, focus on platinum alone, but it is usually only one part of the catalyst recipe.
Palladium and Why It Is Common in Gasoline Applications
Palladium handles many of the same oxidation jobs, yet it has become especially important in gasoline vehicles. PMRCC highlights its strong catalytic activity, corrosion resistance, and solid durability in gasoline exhaust conditions. It also supports faster light-off, which means the converter starts working sooner after startup. So when people wonder how much palladium is in a catalytic converter, the better first question is which vehicle and engine the converter was built for.
Rhodium and Why It Draws So Much Attention
Rhodium stands apart because its main specialty is reducing nitrogen oxides, or NOx, into nitrogen. That matters because NOx control is one of the hardest parts of emissions treatment. The same Phoenix source describes rhodium as the rarest of the three main catalyst metals, which helps explain why even small amounts attract attention in recycling. Questions like how much rhodium is in a catalytic converter matter for the same reason: tiny loadings can still influence recovery value.
| Metal | Primary emissions-control job | Common application tendency | Why recyclers care | Plain-language reason it affects value |
|---|---|---|---|---|
| Platinum | Oxidizes carbon monoxide and hydrocarbons | Important in many diesel systems and some mixed catalyst strategies | It is a recoverable platinum group metal with strong industrial use | Its presence adds value, but the amount varies by converter design |
| Palladium | Also oxidizes carbon monoxide and hydrocarbons | Common in many gasoline passenger vehicles | Often a major part of gasoline converter recovery potential | It is widely used where strong gasoline emissions control is needed |
| Rhodium | Reduces NOx into nitrogen | Critical in systems that need strong nitrogen oxide control | Its scarcity makes it closely watched during assay and pricing | Even thin amounts can matter because rarity increases its importance |
The same three names come up again and again, but the balance is not fixed. A gasoline car, a diesel vehicle, and a larger engine may each need a different catalyst recipe because the emissions target is not exactly the same.
What Metals Are in a Catalytic Converter by Vehicle Type
Here is where the topic gets more specific. The same precious metals come up again and again, but the recipe is not universal. If you have searched what metals are in a catalytic converter, the more useful question is this: in which kind of vehicle? Guidance from Johnson Matthey and technical detail from PMRCC show that catalyst chemistry changes with exhaust conditions, fuel type, and emissions goals, not just with the size of the part.
Gasoline and Diesel Converters Use Different Strategies
Does a diesel engine have a catalytic converter? Yes, but usually not the same kind of unit found on a gasoline passenger car. Most gasoline vehicles use a three-way catalyst that helps treat carbon monoxide, hydrocarbons, and nitrogen oxides in one main converter. Diesel engines run lean, which means there is more oxygen in the exhaust. That changes the chemistry. Instead of relying on a traditional three-way setup, diesel systems often use a diesel oxidation catalyst, a particulate filter, and SCR for NOx control. So when people ask what precious metals in catalytic converters, the first thing to know is that gasoline and diesel exhaust need different solutions.
Vehicle Type, Engine Size, and Emissions Rules Matter
The question what are catalytic converters made of sounds simple, but automakers tune the metal mix around the pollutants a vehicle must control. A small commuter car does not create the same exhaust conditions as a turbocharged SUV, a hybrid, or a heavy-duty diesel truck. That is why what are cat converters made of can mean different metal balances, different substrates, and sometimes extra aftertreatment components.
- Engine type: gasoline, diesel, hybrid, and alternative-fuel engines create different exhaust chemistry.
- Vehicle category: passenger cars, work trucks, and heavy-duty vehicles need different flow capacity and durability.
- Engine size and output: larger or boosted engines can require stronger thermal performance and different catalyst loading.
- Emissions requirements: stricter standards may push designs toward more efficient catalysts or added system stages.
- Converter location: units placed closer to the engine heat up faster, which affects design priorities.
- Intended durability: towing, stop-and-go driving, and repeated cold starts all influence the final build.
Why Older and Newer Units Can Differ
Converter design keeps evolving. Newer gasoline direct-injection vehicles may add particulate control, while hybrids often need catalysts that can handle frequent heat cycling and fast light-off. Diesel systems have also become more modular over time. So when readers ask what are catalytic converters made of, there is no single formula that fits every model year. The metal mix follows the emissions job. That is also why two converters that look similar on the outside may carry very different coatings inside, even when the total amount of precious metal is smaller than most people expect.
How Much Precious Metal Is in a Catalytic Converter?
By this point, the surprising part is not which metals are used, but how little physical material can do the job. If you are searching how much platinum is in a catalytic converter, or how much platinum is inside a catalytic converter, the safest answer is usually less than most people expect. The precious metals inside a catalytic converter are not packed inside as nuggets or thick plates. They are spread as very thin catalytic layers over a large internal surface.
Why the Amount Is Usually Smaller Than People Expect
The internal design explains why. DieselNet describes the washcoat as a porous, high-surface-area layer whose job is to disperse catalytic metals efficiently. PMRCC shows that platinum, palladium, and rhodium sit in that catalyst layer on the honeycomb structure. So if you have asked what is the precious metal inside a catalytic converter, the practical answer is usually a combination of those three metals, but in thin coatings rather than visible chunks.
Catalytic converters work by spreading tiny amounts of precious metal across a huge surface area.
How Thin Coatings Can Still Be Highly Effective
Think of the honeycomb core as a way to create thousands of reaction sites in a compact space. Exhaust gas flows through many small channels, touching coated surfaces again and again. That high surface area is what makes the chemistry effective. It also answers another common question, what precious metal is inside a catalytic converter and why does so little of it matter so much. The catalyst works at the surface, so efficiency matters more than bulk weight.
Why Small Metal Content Can Still Create High Value
What expensive metal is in a catalytic converter? Usually platinum, palladium, and rhodium, with rhodium getting special attention because it is especially rare and important for NOx reduction. The market context from PMRCC also helps explain why small loadings can still carry real value. These metals are difficult to mine, widely used in industry, and subject to price swings. Recovery also takes specialized processing. So when people ask how much platinum is in a catalytic converter, price is only part quantity. It is also about rarity, demand, and how the converter was originally engineered, which is why two similar-looking units are not always valued the same way.

OEM vs Aftermarket Catalytic Converter Differences
One reason similar-looking converters can end up with very different value is origin. An OEM unit is the original converter designed for the vehicle's factory setup. An aftermarket catalytic converter is a replacement made outside that original supply chain. Both can contain the same core precious metals, but fit strategy, compliance target, and metal loading may differ. That is why a used cat converter is never judged by shell size alone.
How OEM and Aftermarket Units Differ
Hott Exhaust describes OEM converters as parts engineered for exact vehicle fit, original performance, and factory emissions specifications. Aftermarket options cover a much wider range. Some are direct-fit replacements. Others are universal designs meant for broader application. Many buyers choose them for lower cost and easier availability, and some are built to meet EPA or CARB requirements for legal emissions compliance.
| Comparison point | OEM converter | Aftermarket converter |
|---|---|---|
| Main goal | Match factory design, fit, and emissions behavior | Provide a replacement that fits budget, availability, and application needs |
| Typical material priorities | Often more vehicle-specific construction and denser precious-metal loading | Can vary by certification level, price point, and intended use |
| Fit approach | Exact model-specific fit | Direct-fit or universal |
| Compliance considerations | Built around original vehicle requirements | Must match applicable federal or CARB rules where required |
| Durability expectations | Usually aimed at long service life and consistent performance | Ranges from budget replacements to higher-spec certified units |
| Scrap value pattern | Often more predictable and often higher | Often lower or less predictable because recoverable metal varies |
Why Metal Loading and Build Choices Are Not Always Equal
Material differences explain a lot. Noble6 and RRCats both note that OEM units typically carry more platinum, palladium, and rhodium, and they are often easier to identify by stamped markings or part numbers. Aftermarket designs vary more in shielding, weld style, substrate choice, and precious-metal loading. That does not make every replacement unit a poor choice. It simply means cat converter material is less uniform across the category.
What These Differences Mean for Performance and Scrap Value
On the vehicle, a properly matched certified replacement can still be a practical repair. In recycling, consistency matters almost as much as chemistry. The value of catalytic converter scrap usually follows recoverable metal content and how confidently the unit can be identified. That is why cat converter metals, serial markings, and build clues matter when a used cat converter is assessed. In plain terms, catalytic converter scrap value comes from what can actually be recovered, not just what the shell looks like. Buyers know that, so the first real question is usually not "OEM or aftermarket?" but "Which one, exactly?"
What Drives Catalytic Converter Scrap Value
By this stage, the chemistry starts turning into a real-world question: how much are catalytic converters worth? The honest answer is that quotes depend on the exact unit, its condition, and the market for platinum, palladium, and rhodium. Material prices matter, but PMRCC makes an important point: value also depends on how much of those metals are actually recoverable from that specific converter. In other words, shell size alone does not tell you much. That is a big part of what makes a catalytic converter valuable in recycling.
Why Some Converters Are Worth More Than Others
Two units can look similar and still quote very differently. An OEM gasoline converter from a stricter-emissions application may carry more recoverable precious metal than an aftermarket replacement. A large diesel unit can look impressive, yet still bring less if its PGM loading is modest. The grading factors highlighted by noble6 and PMRCC are consistent: converter type, precious metal content, age, physical condition, contamination, and current market conditions all influence value. So if you plan to sell catalytic converter units, assume identification comes first and price comes second.
What Recyclers Look at Before Quoting a Price
- Identify the unit. Buyers start with manufacturer stamps, serial numbers, and part codes on the housing. IndexBox notes these markings are the most reliable first clues.
- Classify the converter. OEM, aftermarket, gasoline, diesel, and pre-cat units do not share the same metal mix or value pattern.
- Check condition. Damage, missing material, or contamination from oil or antifreeze can reduce value and complicate grading.
- Use reference tools carefully. Serial-number catalogs and price lists help, but they are still estimates because identical part numbers can age differently in service.
- Judge recoverable metal potential. Serious recyclers do not rely on guesswork alone. For larger lots, processors may sort by type and assay material to get closer to true recovery value.
How Owners Can Prepare for a Legitimate Evaluation
If you are wondering how much can you sell a catalytic converter for, better documentation usually leads to a more accurate answer. Anyone evaluating a catalytic converter for scrap needs enough information to classify it correctly.
- Keep proof of ownership, repair records, or invoices when possible.
- Photograph serial numbers, heat shields, and overall shape before transport.
- Avoid smashing, gutting, or cutting the unit open, since that can destroy identification and lower cat converter scrap value.
- Store the part in a dry place to limit extra damage or contamination.
- Use lawful sales channels and expect documentation checks, since many areas regulate converter transactions closely.
So, how much is a catalytic converter worth in scrap? There is no one-size-fits-all number. The best quote comes from accurate identification, honest condition assessment, and realistic recoverable metal potential at that moment in the market. That same need for exact specs and traceable build details matters long before a converter reaches the recycler, which is why manufacturing precision deserves a closer look.
Using Metal Knowledge in Automotive Manufacturing Decisions
The same detail that affects recycling value also shapes manufacturing decisions up front. If what makes catalytic converters valuable is a carefully engineered mix of platinum, palladium, and rhodium, the surrounding hardware still has to protect that catalyst, manage heat, and hold alignment under real driving loads. That matters for buyers sourcing converter shells, flanges, brackets, bushings, and other exhaust-side components tied to the broader catalytic converter function.
Why Precision Around Emissions Parts Matters
Ask, what is the function of a catalytic converter, and the simple answer is to help convert harmful exhaust gases into less harmful ones. That chemistry depends on more than the catalyst alone. Poor fit, unstable material quality, or inconsistent machining in nearby assemblies can create leakage, vibration, or thermal stress. For engineering and sourcing teams, understanding what are the precious metals in catalytic converters helps clarify why these systems are expensive and tightly controlled. It also answers part of the question, what is in catalytic converter that makes it valuable: rare catalyst metals, plus the precision needed to keep the whole assembly working reliably.
What Automotive Buyers Should Look for in a Manufacturing Partner
In the automotive supply chain, IATF 16949 matters because it supports defect prevention, continuous improvement, and the use of core tools such as SPC. For prototype work, rapid prototyping also helps teams test and refine parts faster before locking in production.
- Process control: Look for disciplined monitoring methods such as SPC on critical dimensions.
- Material consistency: Stable incoming material and repeatable processing reduce variation.
- Prototype responsiveness: Fast sample turnaround helps validate fit, heat exposure, and assembly logic early.
- Production scalability: The supplier should support one-off prototypes, pilot runs, and automated volume output.
A Practical Next Step from Prototype to Volume
If your team is sourcing precision-machined metal parts for emissions-system assemblies rather than catalyst chemistry itself, Shaoyi Metal Technology is one relevant resource to review. Its published capabilities include IATF 16949-certified automotive machining, SPC-based process control, rapid prototyping, and mass production support, with the company stating it serves 30+ automotive brands worldwide. For buyers weighing why are catalytic converters so expensive, that broader picture matters: rare metals drive core value, but manufacturing discipline is what turns a design into a repeatable automotive part.
Common Questions About Catalytic Converter Metals
1. What precious metals are in a catalytic converter?
Most catalytic converters use platinum, palladium, and rhodium. These metals are spread across an active coating on the inner honeycomb core, where they speed up reactions that reduce harmful exhaust emissions. The exact mix depends on the vehicle, engine type, and emissions strategy.
2. How much platinum is inside a catalytic converter?
Usually far less than many people expect. The metal is applied as a very thin layer over a large internal surface, so the converter works through surface contact rather than heavy pieces of platinum. That is why even a small amount can still matter for manufacturing cost and recycling value.
3. Are all catalytic converters made with the same metal mix?
No. Gasoline and diesel systems need different chemical approaches, and design choices also change with vehicle size, model year, emissions rules, and durability targets. Two converters can look similar from the outside while using very different proportions of platinum, palladium, and rhodium inside.
4. Why are some catalytic converters worth more for scrap?
Scrap value depends on the exact converter, not just its size or weight. Buyers usually check the part number, whether it is OEM or aftermarket, its physical condition, and the likely recoverable amount of platinum group metals. Current metal markets affect quotes too, but correct identification is often the starting point.
5. What should manufacturers look for in a partner for emissions-system metal parts?
For converter housings, flanges, brackets, and related exhaust hardware, buyers should focus on stable materials, tight process control, prototype speed, and the ability to scale into production. Quality systems such as IATF 16949 and tools like SPC help reduce variation and improve consistency. A supplier such as Shaoyi Metal Technology can be a useful reference for teams that need precision machining support for adjacent emissions-system parts from prototype to volume production.
 Small batches, high standards. Our rapid prototyping service makes validation faster and easier —
Small batches, high standards. Our rapid prototyping service makes validation faster and easier — 
