What Is the Metal in a Catalytic Converter? Inside the Precious Mix

The Short Answer on Catalytic Converter Metals
If you are asking what is the metal in a catalytic converter, the most accurate answer is not one metal, but several. In most modern units, the active catalyst is a mix of platinum-group metals, mainly platinum, palladium, and rhodium, applied as a thin coating on an internal substrate. The outer housing, by contrast, is commonly stainless steel. So what is in a catalytic converter depends on whether you mean the shell or the catalyst itself.
A catalytic converter usually contains platinum, palladium, and rhodium on an internal substrate, while the outside case is typically stainless steel.
What Is the Metal in a Catalytic Converter
People often ask what is the precious metal inside a catalytic converter as if there is a single answer. Sources from IPA and PMR show that the catalyst layer typically uses a combination of platinum, palladium, and rhodium because those metals help convert harmful exhaust gases into less harmful ones. If you have wondered what's in a catalytic converter, the key is separating the chemical metals from the structural parts.
Why Catalytic Converter Metal Means More Than One Metal
- The valuable catalyst is usually a blend of platinum, palladium, and rhodium, not one standalone metal.
- Those metals are spread across a honeycomb-like internal surface, not stored as visible chunks.
- The part you can see from outside is generally a stainless steel canister that protects the active materials.
Stainless Steel Shell Versus Precious Metal Coating
This is where many quick answers go wrong. If someone asks what is the metal inside a catalytic converter, they may mean the stainless steel shell, or they may mean the precious catalyst coating inside. Both are real parts of the assembly, but they do different jobs. The shell handles heat and protection. The platinum-group metals handle the chemistry. That simple distinction opens the door to a more useful question: what is actually layered inside the converter, and where do those metals sit?
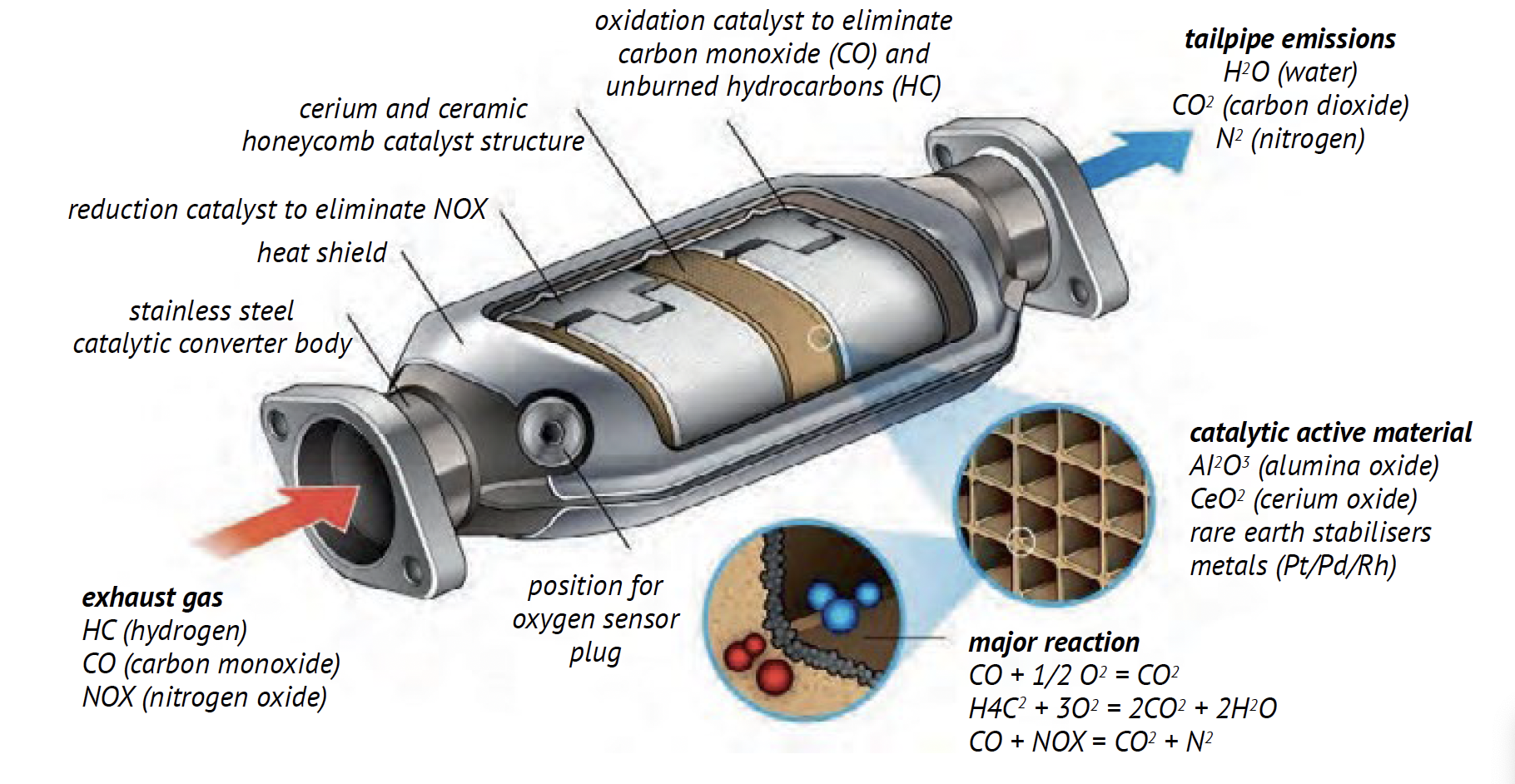
Catalytic Converter Inside
That shell-versus-catalyst distinction makes more sense when you picture the unit as a set of engineered layers. If you imagine the inside of a catalytic converter as a chamber full of metal pieces, the real design is much smarter. The catalytic converter inside is usually a stainless steel canister protecting a honeycomb core, and the precious metals sit on that core as ultra-thin coatings rather than loose bits.
What Is Inside a Cat Converter
When people search for cat converter inside diagrams, they are usually trying to understand the assembly from the outside in. A typical converter includes:
- Stainless steel housing: the outer shell that handles heat, corrosion, and mounting.
- Support mat: a cushioning and sealing layer that holds the core in place and helps absorb vibration and thermal expansion.
- Substrate: the internal ceramic or metallic monolith shaped like a honeycomb.
- Washcoat: a porous coating on the honeycomb walls that greatly increases reactive surface area.
- Catalyst metals: platinum, palladium, and rhodium dispersed across the washcoat.
This layered structure is consistently described by Jendamark, Catman, and AECC.
How the Honeycomb Substrate Holds Catalytic Materials
The substrate is the working core. It is usually ceramic or metal, and its honeycomb shape lets exhaust flow through many narrow channels. That creates a very large surface area in a compact part. More surface area means more contact between hot exhaust gases and the catalyst coating. AECC also notes that modern substrates can use thin walls and high cell density, which helps efficiency and quicker warm-up.
Where the Active Metals Sit Inside the Converter
The active metals are not stored as visible chunks inside of a catalytic converter. They are spread as a thin catalytic layer on the washcoat that covers the channel walls. In simple terms, the honeycomb supplies thousands of tiny pathways, and the washcoat gives those pathways a rough, porous skin. The metals are distributed across that skin so passing exhaust can touch them over and over again.
For readers looking for inside catalytic converter details, that point matters most: the chemistry depends on placement, not just metal names. Two units can look similar on the outside while behaving differently inside. The reason comes down to the specific roles of platinum, palladium, and rhodium.
Platinum, Palladium, and Rhodium Compared
The honeycomb structure explains where the catalyst sits. The next question is what the catalyst actually is. When people ask what metal is in catalytic converters, they are usually asking about the active metals that do the exhaust-cleaning work. In a modern three-way converter, that usually means platinum, palladium, and rhodium, each handling a different part of the chemistry rather than acting as interchangeable names.
Platinum Palladium and Rhodium at a Glance
| Metal | Main catalytic role | Why it is used | How it differs | Where it is emphasized |
|---|---|---|---|---|
| Platinum | Oxidation catalyst for CO and HC | Helps convert harmful carbon monoxide and unburned hydrocarbons into less harmful gases | Shares the oxidation job with palladium instead of handling NOx reduction | Oxidation side of a three-way converter |
| Palladium | Oxidation catalyst for CO and HC | Supports the same broad cleanup reactions as platinum | Usually discussed with platinum because both serve the oxidation function | Oxidation side of a three-way converter |
| Rhodium | Reduction catalyst for NOx | Helps convert nitrogen oxides into nitrogen and oxygen | Handles reduction, which is the opposite reaction from platinum and palladium | Reduction section, typically positioned first |
What Each Precious Metal Does in Exhaust Treatment
This division of labor is the real answer behind searches like what precious metals are in a catalytic converter. Materials on precious metals show that platinum and palladium primarily drive oxidation reactions, turning CO and HC into CO2 and H2O. Rhodium is crucial for reduction, helping convert NOx into N2 and O2. Another breakdown of reduction and oxidation catalysts notes that rhodium is typically associated with the first reduction stage, while platinum and palladium support the oxidation stage that follows.
If you are comparing catalytic converter platinum with palladium, the key shared point is oxidation. If you are asking what is rhodium used for, its standout job is NOx reduction. People searching what precious metals in catalytic converters actually do usually want that simple map.
Why Rhodium Is Important but Not the Only Valuable Metal
Rhodium often gets extra attention in discussions of rare converter metals, but no single precious metal in catalytic converter chemistry does everything. Rhodium is vital because NOx reduction is a separate task. Still, platinum and palladium remain central to overall converter performance because the unit also has to oxidize carbon monoxide and hydrocarbons. In plain English, the converter works as a coordinated system, not a one-metal device. That is why two converters can share the same three metal names on paper and still use different balances in practice.
Why Catalytic Converter Metals Change by Vehicle
The same three metal names do not always show up in the same proportions. That is why one converter may lean more on palladium, another may favor platinum, and another may use a different balance of all three. If you are asking what is a catalytic converter made of, the useful answer is tied to engine behavior, emissions goals, heat, and packaging, not just a fixed recipe.
Why the Metal Mix Changes by Vehicle
Searches for what are cat converters made of often assume every unit follows one universal formula. In practice, automakers tune the catalyst mix around the vehicle it serves. Guidance from PMRCC shows that engine type, oxygen levels in the exhaust, system layout, and durability demands all shape converter design. Metal price swings also matter, because manufacturers may rebalance platinum and palladium without giving up emissions performance.
- Engine type: gasoline and diesel exhaust have different chemistry.
- Emissions strategy: the system must target CO, hydrocarbons, NOx, and sometimes particulates in different ways.
- Temperature target: the catalyst has to heat up quickly and keep working under load.
- Converter position: a unit near the engine sees hotter gas than one mounted farther downstream.
- Packaging and size: engine layout, turbo hardware, and available space affect substrate design and catalyst loading.
- Material strategy: automakers adjust the precious-metal balance as supply and cost change.
Gasoline Diesel and Design Differences
Gasoline engines usually run close to stoichiometric conditions, which lets a three-way catalyst handle oxidation and reduction in the same system. PMRCC notes that these converters typically use platinum, palladium, and rhodium, with rhodium especially important for NOx reduction and palladium commonly emphasized in many modern gasoline designs. Diesel is a different story. Lean-burn diesel exhaust contains excess oxygen, so it often relies on a modular setup such as a diesel oxidation catalyst, a particulate filter, and SCR or a lean NOx trap. So, does a diesel engine have a catalytic converter? Yes, but often as part of a broader aftertreatment system rather than a single gasoline-style three-way unit. Recohub similarly notes that diesel units often rely primarily on platinum and palladium.
Why Two Converters Can Look Similar but Contain Different Metals
Outside appearance can be misleading. Two stainless canisters may look almost identical, yet one may be placed close to the manifold for faster light-off, while the other sits farther down the exhaust stream and runs cooler. A short explainer on close-coupled placement highlights why that matters: hotter exhaust helps the catalyst reach operating temperature faster, especially during cold starts.
Exact platinum, palladium, and rhodium loadings cannot be confirmed reliably without model-specific records or lab analysis.
That is why what are catalytic converters made of has more than one valid answer across the market. The shell may look familiar, but the chemistry inside depends on fuel type, exhaust temperature, placement, and compliance targets. Even then, one practical mystery remains: the actual amount of each precious metal is usually much smaller, and much harder to judge, than most people expect.

How Much Precious Metal Is Actually Inside
People often ask how much platinum is in a catalytic converter, how much palladium is in a catalytic converter, or how much rhodium is in a catalytic converter as if there is one standard number. There is not. These metals are usually present in small amounts and spread as thin catalytic coatings across the washcoat on the honeycomb substrate, not sitting inside as visible chunks. That is why quantity questions need a careful answer. The loading can change a lot by vehicle model, engine size, fuel type, converter position, and emissions package.
How Much Platinum Palladium and Rhodium Can Be Present
Reliable published figures are usually broad, not exact for every car. Thermo Fisher notes that the recoverable platinum, palladium, and rhodium together can range from about 1 to 2 grams for a small car to roughly 12 to 15 grams for a big truck in the U.S. That is a combined total, not a per-metal promise. For rhodium specifically, PMRCC explains that most gasoline vehicles contain only fractions of a gram, though newer models may use higher rhodium loadings to satisfy tighter emissions rules. So if you are wondering how much platinum is inside a catalytic converter, the honest answer is always model dependent.
| General Patterns | Model-Specific Unknowns |
|---|---|
| Precious metals are usually present as thin coatings, not solid pieces | The exact grams of platinum, palladium, and rhodium in one converter |
| Rhodium in gasoline vehicles is often only a fraction of a gram | The precise Pt-Pd-Rh ratio used for a given engine and emissions certification |
| Combined recoverable PGM content can vary widely across vehicle classes | Whether a specific unit is platinum-heavy, palladium-heavy, or uses a different balance |
| Outside size does not reliably show metal loading | True content usually requires part-number data or lab analysis |
Why Small Amounts of Precious Metal Still Matter
Small does not mean unimportant. The coating is spread over a huge internal surface area, so even tiny amounts can contact a large volume of exhaust gas and drive the needed reactions. That is why searches like how much rhodium is inside a catalytic converter matter even when the answer may sound modest. A fraction of a gram can still be chemically essential, especially for NOx reduction, and the same logic applies to platinum and palladium.
What Cannot Be Known by Visual Inspection Alone
You cannot look at the shell, shake the unit, or compare canister size and know the true metal content. Two converters may appear similar but carry very different loadings. Even experienced recyclers rely on part identification and analytical methods because how much rhodium is inside a catalytic converter cannot be confirmed by sight alone. That hidden, thinly distributed metal is also a big reason a seemingly ordinary converter can carry surprisingly high material value.
Why Are Catalytic Converters So Expensive?
The tiny coating on the honeycomb helps explain the big price tag. People asking why are catalytic converters so expensive are really comparing two things: the value of the precious metals inside and the full cost of a compliant replacement part. Those numbers overlap, but they are not identical. Platinum, palladium, and rhodium do the emissions work, and all three trade in volatile global markets. So, are catalytic converters expensive? Often yes, but not just because they contain valuable metal.
Why Catalytic Converters Are Expensive
A practical answer to why is the catalytic converter so expensive starts with rarity and function. PMR notes that about 60% of global platinum-group metal production goes into catalytic converters, where those metals must withstand heat, corrosion, acids, and constant exhaust flow. RRCats also shows how sensitive pricing can be: a $100 per ounce move in rhodium, platinum, or palladium can change a converter quote by tens of dollars.
- Rare metals: platinum-group metals are scarce, and rhodium is especially rare.
- Market volatility: mining output, trade shifts, and supply disruptions can move prices quickly.
- Emissions compliance: the converter is a regulated, engineered part, not just a metal canister.
- Replacement realities: manufacturing, shipping, sourcing, and labor add cost beyond raw metal value.
How Precious Metal Content Influences Value
When people ask how much are catalytic converters worth, it helps to separate scrap value from replacement cost. Scrap value depends on the catalytic converter value metal mix, current PGM prices, and unit type. PMR explains that aftermarket converters typically contain about 10% of the PGM content found in OEM units, so two similar-looking parts may have very different recycling value. Replacement cost is broader. It can also reflect manufacturing, shipping, supply pressure, and labor. On Miller CAT, a reported example showed an OEM Prius converter list price rising from about $2,466 to $3,038 in ten months.
Why Rhodium Gets So Much Attention
If you are wondering what expensive metal is in a catalytic converter, rhodium usually gets the headlines. PMR describes it as especially rare and mainly recovered as a by-product, while RRCats calls it the most volatile and valuable of the three key metals, often exceeding $10,000 per ounce in recent years. Still, the catalytic converter value metal story is not only rhodium. Platinum and palladium remain central to converter performance and real-world value as well.
That is why headlines alone cannot tell you what a specific unit is worth. Actual value depends on verified content, unit type, and condition, not just one market chart. Since the shell tells only part of the story, outside clues and part identification matter far more than many owners expect.
Where Is the Catalytic Converter Located on a Car?
Material value gets attention, but identification starts with the outside of the vehicle. If you are wondering where is the catalytic converter located, the usual answer is in the exhaust system between the engine and the muffler or mufflers. A guide from CarParts notes that some vehicles have a pre-cat near or built into the exhaust manifold and a main cat farther downstream. So when people ask how many catalytic converters in a car, the real answer can be one or several, depending on engine layout and emissions design.
Where the Catalytic Converter Is Located
To locate catalytic converter placement safely, trace the exhaust path rather than guessing from a random heat shield. On V-shaped or flat engines, there may be converters on each bank, and some vehicles can have up to four. Repair information may also label them as bank 1 or bank 2. If you are asking what does a catalytic converter look like, look for a metal-cased section in the exhaust assembly, but remember that external shape alone is not enough to identify its internal metal mix.
How to Read External Clues Before Assuming Metal Content
- Check vehicle-specific information first. A service manual or repair database is the safest way to confirm location and application.
- Follow the exhaust line visually. Look between the engine and muffler area for the converter or converters.
- Read external markings only. Part numbers, serial numbers, bank labels, and flow-direction marks are more useful than appearance alone.
- Note aftermarket clues. RRCats points to common signs such as a silver shield with an arrow, stamps like "Flow" or "Out," and some serial numbers beginning with "N."
- Stop at external inspection. Do not remove, cut, or open the unit to guess what is inside.
Why OEM and Aftermarket Units Can Differ
An aftermarket catalytic converter may be easier to spot from those outside clues, but that still does not tell you the exact platinum, palladium, or rhodium loading. RRCats notes that aftermarket units often contain less precious metal than OEM parts, yet the amount varies by application. Not all converters have visible numbers, and two units can look similar while serving different vehicles or emissions standards. That is why serial markings, vehicle fitment, and documented application matter more than a quick glance under the car. External identification tells you what the part likely is. Determining how well it fits, seals, and performs brings in another layer entirely: the precision of the surrounding exhaust hardware.

Choosing Reliable Metal Fabrication Support for Exhaust Components
The precious coating answers the chemistry question, but the surrounding hardware decides whether the unit fits, seals, and survives. In an automobile catalytic converter, the outer can, pipework, flanges, brackets, and sensor bosses all need tight dimensional control. BM Catalysts notes that converter cans and pipe sections are commonly made from 409 grade stainless steel because it offers strength, corrosion resistance, and the formability needed for exhaust parts. That is a useful reminder that the catalytic converter metal people talk about most is only one part of the total build.
Why Precision Matters Around Catalytic Converter Assemblies
Ask what is the function of a catalytic converter in real service, and the answer goes beyond chemistry. The assembly has to keep exhaust moving through the substrate, hold the monolith securely, manage heat expansion, and keep sensors in the right position. BM Catalysts also describes fitment parts such as flanges, lambda bosses, and brackets as separately manufactured pieces, because each one has its own tolerance and joining demands. So when buyers focus on catalytic converter metals, they should also evaluate the catalytic converter material used in the housing and support hardware.
From Prototype to Mass Production for Automotive Metal Parts
For procurement teams, repeatability is the real test. Smithers describes IATF 16949 as the automotive quality framework built around continuous improvement, defect prevention, and core tools such as SPC and PPAP. That matters for exhaust hardware because prototype parts, trial builds, and production runs should all follow the same quality logic. One manufacturing resource worth reviewing is Shaoyi Metal Technology, which presents IATF 16949-certified automotive machining, SPC-based control, and support that ranges from rapid prototyping to automated mass production for metal components around exhaust assemblies.
What to Look for in an Automotive Machining Partner
- Experience with housings, flanges, brackets, sensor bosses, and pipe sections used near exhaust heat.
- Automotive quality systems aligned with IATF 16949.
- Process control on critical dimensions, not only final inspection.
- Capacity to move from one prototype to production volume without losing traceability.
- Material knowledge for stainless steel and other grades used near corrosion and thermal cycling.
- Clear drawing review, inspection reporting, and communication with sourcing teams.
That checklist matters because metal in catalytic converters is only valuable when the surrounding structure lets it work reliably. In manufacturing terms, catalytic converter metal is not just about platinum-group chemistry. It is also about whether the supporting metalwork is precise enough to protect that chemistry on the road.
FAQ: Catalytic Converter Metals
1. What precious metals are in a catalytic converter?
Most modern catalytic converters use platinum-group metals, mainly platinum, palladium, and rhodium. These are not packed inside as solid pieces. Instead, they are spread as a very thin active layer over a honeycomb substrate so exhaust gas can touch a large reactive surface. Platinum and palladium are commonly associated with oxidation reactions, while rhodium is especially important for reducing nitrogen oxides. The exact mix changes by vehicle, engine type, emissions rules, and converter design.
2. Is the outer shell of a catalytic converter made of the same metal as the catalyst?
No. The visible outer case is usually stainless steel because it needs strength, heat resistance, and corrosion protection. The valuable catalytic metals are inside the unit on the coated substrate. This is why the question can be confusing: one answer refers to the structural housing, while the other refers to the precious metals that actually clean the exhaust. In simple terms, the shell protects the part, and the platinum-group metals perform the chemical reactions.
3. How much rhodium is inside a catalytic converter?
Usually far less than many people assume. Rhodium is often present in very small amounts, sometimes only fractions of a gram in many gasoline applications, yet it still plays a major role because it is highly effective in NOx reduction. The real amount depends on the vehicle model, engine size, emissions package, and converter location in the exhaust system. You cannot confirm rhodium content by sight alone. Reliable identification usually requires part-number data or analytical testing.
4. Do diesel catalytic converters use the same metal mix as gasoline converters?
Not always. Gasoline vehicles often use a three-way catalyst that combines oxidation and reduction functions in one emissions-control setup, commonly involving platinum, palladium, and rhodium. Diesel exhaust works under different conditions, especially because it usually contains excess oxygen, so diesel aftertreatment systems are often more modular. They may use different balances of platinum-group metals and work alongside parts such as diesel oxidation catalysts, particulate filters, or SCR systems. So the metal strategy can differ even when the units look similar from outside.
5. Why does precision metal fabrication matter for catalytic-converter-related parts?
Catalyst chemistry gets the attention, but the surrounding metal parts determine whether the system fits, seals, and survives real operating conditions. Housings, flanges, brackets, pipe sections, and sensor bosses all need tight tolerances so exhaust flow, heat expansion, and sensor placement stay under control. For automotive manufacturers, quality systems such as IATF 16949 and process methods like SPC help keep those parts consistent from prototype to mass production. That is why sourcing teams may review suppliers such as Shaoyi Metal Technology when evaluating machining support for exhaust-adjacent components.
 Small batches, high standards. Our rapid prototyping service makes validation faster and easier —
Small batches, high standards. Our rapid prototyping service makes validation faster and easier — 
