โลหะดินอัลคาไลน์คืออะไร? กลุ่มที่ 2 ตอนนี้เข้าใจได้ชัดเจนแล้ว
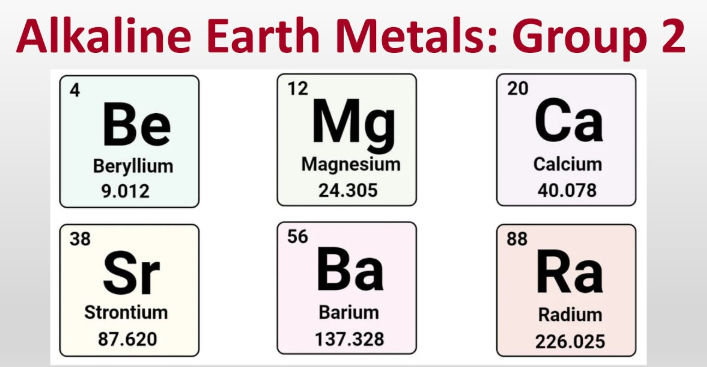
โลหะดินกลุ่มอัลคาไลน์คืออะไร
หากคุณค้นหาคำว่า โลหะดินกลุ่มอัลคาไลน์คืออะไร ที่นี่คือคำตอบโดยตรง: คือธาตุทั้งหกชนิดใน กลุ่ม 2 ของตารางธาตุ คำนิยามสั้นๆ เรื่อง โลหะดินกลุ่มอัลคาไลน์ นี้เป็นจุดเริ่มต้น แต่ชื่อก็บ่งบอกพฤติกรรมของธาตุกลุ่มนี้ได้มากเช่นกัน
โลหะดินกลุ่มอัลคาไลน์คืออะไร ในหนึ่งประโยค
โลหะดินกลุ่มอัลคาไลน์ ได้แก่ เบริลเลียม แมกนีเซียม แคลเซียม สตรอนเทียม บาริอุม และเรเดียม ซึ่งเป็นธาตุโลหะทั้งหกชนิดในหมู่ที่ 2 ที่มักจะสร้างไอออนประจุ +2
- เบริลเลียม (Be)
- แมกนีเซียม (Mg)
- แคลเซียม (Ca)
- สตรอนเทียม (Sr)
- บาริอุม (Ba)
- เรเดียม (Ra)
เหตุใดชื่อ 'โลหะดินกลุ่มด่าง' จึงสอดคล้องกับลักษณะของธาตุเหล่านี้
สำหรับผู้เริ่มต้น นิยามของโลหะดินกลุ่มด่าง จะเข้าใจได้ง่ายขึ้นมากเมื่อคุณแบ่งวลีนี้ออกเป็นสามส่วน
ด่าง หมายความว่า ออกไซด์และไฮดรอกไซด์ของธาตุเหล่านี้มีลักษณะเป็นเบส ไม่ใช่กรด โลก เป็นคำที่มีมาแต่โบราณ นักเคมีในยุคแรกใช้คำนี้เพื่ออธิบายสารที่ทนความร้อนได้ดี มีลักษณะคล้ายแร่ธาตุ และไม่หลอมละลายหรือละลายในน้ำได้ง่าย ซึ่งจุดนี้อธิบายไว้โดย บริตันนิกา . โลหะ หมายความว่า ธาตุเหล่านี้เองมีลักษณะเป็นโลหะ โดยทั่วไปมีผิวมันวาว และสามารถสูญเสียอิเล็กตรอนได้ดีในการทำปฏิกิริยา
สิ่งนั้นจะให้คุณได้รับความเข้าใจพื้นฐานเกี่ยวกับ ความหมายของโลหะดินกลุ่มด่าง โดยไม่จำเป็นต้องเข้าใจเคมีขั้นสูงก่อน ทั้งนี้ยังเป็นคำอธิบายสั้นๆ ที่มีประโยชน์ เกี่ยวกับโลหะแอลคาไลน์เอิร์ธ : กลุ่มโลหะหมู่ที่ 2 ซึ่งมีสมบัติทางเคมีร่วมกัน มีตำแหน่งร่วมกันบนตารางธาตุ และมีบทบาทสำคัญในโลกแห่งความเป็นจริง แมกนีเซียมพบได้ในโลหะผสมและสิ่งมีชีวิต แคลเซียมมีความสำคัญต่อโครงสร้างกระดูก เปลือกหอย และวัสดุก่อสร้าง ส่วนแบริอุม สตรอนเทียม และเรเดียมปรากฏในบริบทที่เฉพาะเจาะจงมากขึ้น
บทความนี้เริ่มต้นด้วยเนื้อหาพื้นฐานโดยตั้งใจ เพื่อให้ผู้อ่านสามารถท่องจำรายการที่เรียบง่ายได้ง่าย แต่การเข้าใจ 'ครอบครัว' ของธาตุเหล่านี้อย่างแท้จริงจะเกิดขึ้นเมื่อคุณเห็นตำแหน่งที่ธาตุเหล่านี้ตั้งอยู่บนตารางธาตุ และเข้าใจว่าเหตุใดตำแหน่งนั้นจึงทำให้ธาตุเหล่านี้มีพฤติกรรมที่คล้ายคลึงกันมาก
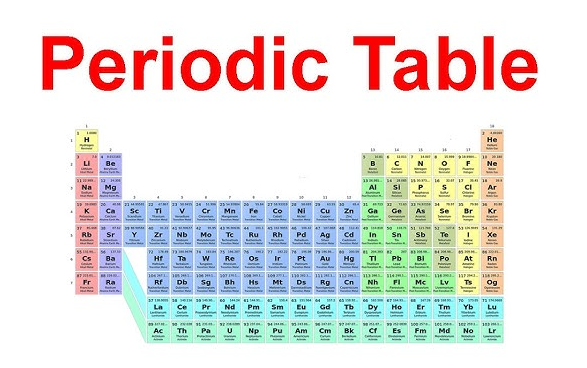
ตำแหน่งของหมู่ที่ 2 บนตารางธาตุ
ชื่อของกลุ่มธาตุจะจดจำได้ง่ายขึ้นมาก หากคุณสามารถระบุตำแหน่งของมันได้จริง ธาตุแอลคาไลน์เอิร์ธอยู่ที่ใดบนตารางธาตุ ให้ดูที่คอลัมน์ที่สองจากด้านซ้าย คอลัมน์แนวตั้งนี้คือ หมู่ที่ 2 บนตารางธาตุ ซึ่งตั้งอยู่ทันทีถัดจากโลหะแอลคาไลในหมู่ที่ 1 กลุ่ม 2 ของตารางธาตุ ภาพแสดงเส้นตรงเดียวกันทุกครั้ง: เบอริลเลียมอยู่ด้านบนสุด ตามด้วยแมกนีเซียม แคลเซียม สตรอนเทียม แบเรียม และเรเดียม ซึ่งจัดเรียงลงมาตามคาบต่างๆ
ในอุโมงค์ ตารางธาตุของโลหะอัลคาไลน์เอิร์ธ แผนภูมิ ธาตุทั้งหกชนิดนี้จัดอยู่ในบล็อก s การที่ธาตุเหล่านี้อยู่ในตำแหน่งเดียวกันมีความสำคัญ เพราะสะท้อนรูปแบบการจัดเรียงอิเล็กตรอนที่เหมือนกัน LibreTexts อธิบายว่า ธาตุในกลุ่ม 2 มีการจัดเรียงอิเล็กตรอนเวเลนซ์เป็น ns 2ซึ่งหมายความว่าธาตุเหล่านี้มีอิเล็กตรอนสองตัวในเปลือกนอกสุด
ตำแหน่งที่พบโลหะอัลคาไลน์เอิร์ธบนตารางธาตุ
โดยสายตา รูปแบบนี้เรียบง่ายมาก ธาตุในกลุ่ม 2 ของตารางธาตุ จัดเป็นคอลัมน์หนึ่งครอบคลุมคาบ 2 ถึง 7 แผนผังการสอนหลายแบบในห้องเรียนเน้นให้เห็นคอลัมน์นี้อย่างชัดเจน โลหะอัลคาไลน์เอิร์ธในตารางธาตุ รูปแบบการจัดวางที่มีสีเดียวกัน เนื่องจากครอบครัวของธาตุถูกอ่านตามแนวตั้ง ไม่ใช่แนวนอน การค้นหาด้วยคำว่า ตารางธาตุ โลหะอัลคาไลน์เอิร์ธ แท้จริงแล้วหมายถึงคอลัมน์นั้นคอลัมน์เดียว
| ธาตุ | สัญลักษณ์ | ตำแหน่งกลุ่มที่ 2 | ไอออนทั่วไป | สารประกอบที่คุ้นเคย |
|---|---|---|---|---|
| เบริลลียม | Be | คาบ 2 ด้านบนของกลุ่มที่ 2 | Be2+ | BeO |
| แมกนีเซียม | Mg | คาบ 3 | Mg2+ | MgO |
| แคลเซียม | Ca | คาบ 4 | Ca2+ | คาโก 3 |
| สโตรนเทียม | Sr | คาบ 5 | Sr2+ | SrCO 3 |
| บารีียม | BA | คาบ 6 | BA 2+ | BaSO 4 |
| เรเดียม | RA | คาบ 7 ด้านล่างของหมู่ที่ 2 | RA 2+ | RaCl 2 |
เหตุใดธาตุในหมู่ที่ 2 จึงสร้างไอออนประจุบวกสองหน่วย
อิเล็กตรอนสองตัวที่อยู่ชั้นนอกสุดเป็นตัวกำหนดปฏิกิริยาเคมี อะตอมของธาตุในหมู่ที่ 2 มักจะสูญเสียอิเล็กตรอนทั้งสองตัว เนื่องจากการสูญเสียนั้นทำให้เกิดการจัดเรียงอิเล็กตรอนที่มีเสถียรภาพมากขึ้น ผลที่ได้คือ +2 ไอออน เช่น Mg 2+ หรือ Ca 2+ นั่นคือเหตุผลที่โลหะเหล่านี้มักสร้างสารประกอบต่าง ๆ เช่น ออกไซด์ คลอไรด์ คาร์บอเนต และซัลเฟต คุณจะสังเกตรูปแบบในสูตรเคมีได้ทันที: MgO, CaCl 2, CaCO 3, BaSO 4.
วิธีระบุธาตุหมู่เอิร์ธแอลคาไลน์อย่างรวดเร็ว
เทคนิคการระบุอย่างรวดเร็วคือการมองหาเบาะแสสามประการพร้อมกัน: อยู่ในคอลัมน์ที่สอง เป็นธาตุโลหะ และมีประจุโดยทั่วไปเป็น +2 เมื่อเปรียบเทียบกับธาตุหมู่แอลคาไลที่อยู่ข้างเคียง ซึ่งมีอิเล็กตรอนวาเลนซ์หนึ่งตัวและมักสร้างไอออนประจุ +1 แล้ว ธาตุในหมู่ที่ 2 จะมีอิเล็กตรอนนอกสุดเพิ่มอีกหนึ่งตัว จึงมีปฏิกิริยาน้อยกว่าโดยทั่วไป ถึงกระนั้น ธาตุเหล่านี้ก็ชัดเจนว่าจัดอยู่ในครอบครัวเดียวกัน ส่วนที่น่าสนใจคือ แต่ละธาตุแสดงรูปแบบนี้ออกมาแตกต่างกันเล็กน้อย โดยเฉพาะจากเบริลเลียมที่อยู่บนสุด ไปจนถึงเรเดียมที่อยู่ล่างสุด
พบกับธาตุเอิร์ธแอลคาไลน์ทั้งหกชนิด
รายการนั้นเป็นประโยชน์ แต่ก็ไม่ค่อยจดจำได้ง่ายนักหากพิจารณาเพียงลำพัง กลุ่มที่ 2 เริ่มรู้สึกมีชีวิตชีวาขึ้นเมื่อสมาชิกแต่ละคนมีเอกลักษณ์ที่ชัดเจน บางตัวพบได้ในกระดูกหรือน้ำทะเล บางตัวให้สีแก่ดอกไม้ไฟ ส่วนอีกตัวหนึ่งส่วนใหญ่แล้วทำหน้าที่เป็นสัญญาณเตือนทางประวัติศาสตร์ แม้จะอยู่ร่วมกันในครอบครัวเดียวกัน แต่แต่ละตัวก็มีบุคลิกภาพของตนเอง
| ธาตุ | สัญลักษณ์ | การเกิดขึ้นทั่วไป | คุณสมบัติโดดเด่น | ความเกี่ยวข้องในโลกแห่งความเป็นจริง |
|---|---|---|---|---|
| เบริลลียม | Be | พบในแร่ธาตุ เช่น เบอริล | มีน้ำหนักเบาและแข็งแกร่งมากสำหรับโลหะ | ใช้ในงานเฉพาะทางด้านการบินอวกาศและการถ่ายภาพรังสีเอ็กซ์; ฝุ่นของมันเป็นอันตรายหากสูดดมเข้าไป |
| แมกนีเซียม | Mg | มีอยู่ในน้ำทะเลและแร่ธาตุ | มีความหนาแน่นต่ำ และให้เปลวไฟสีขาวสดใสเมื่อเผาไหม้ | มีความสำคัญในโลหะผสมน้ำหนักเบา สารเสริม และกระบวนการทางชีววิทยา |
| แคลเซียม | Ca | พบได้ทั่วไปในหินปูน กระดูก เปลือกหอย และชอล์ก | ไอออนกลุ่มที่ 2 ซึ่งมีความคุ้นเคยทางชีวภาพ | มีบทบาทสำคัญในโครงร่างกระดูก ปูนซีเมนต์ ปูนปลาสเตอร์ และแร่ธรรมชาติหลายชนิด |
| สโตรนเทียม | Sr | พบได้เป็นหลักในแร่เซเลสไตรต์ (celestite) และแร่สตรอนเทียนไลต์ (strontianite) | เกลือของธาตุนี้ให้สีแดงสดใสเมื่อเผาในเปลวไฟ | ใช้ในพลุ แสงสัญญาณเตือน (flares) วัสดุเรืองแสง และผลิตภัณฑ์ทันตกรรมบางชนิด |
| บารีียม | BA | มักพบร่วมกับแร่บาริไทต์ (barite) | เป็นโลหะดินกลุ่มด่างที่มีความหนาแน่นสูงและมีน้ำหนักมาก | บาริเยมซัลเฟตมีความสำคัญต่อการขุดเจาะน้ำมันและการถ่ายภาพทางการแพทย์; ขณะที่สารประกอบบาริเยมที่ละลายน้ำได้จำเป็นต้องใช้ด้วยความระมัดระวัง |
| เรเดียม | RA | เกิดขึ้นในปริมาณน้อยมากในแร่ยูเรเนียม | กัมมันตภาพรังสีที่รุนแรงเป็นปัจจัยหลักที่กำหนดพฤติกรรมทางเคมีของธาตุนี้ | ส่วนใหญ่มีความเกี่ยวข้องเชิงประวัติศาสตร์หรือมีความสำคัญทางวิทยาศาสตร์ที่ถูกควบคุมอย่างเข้มงวดในปัจจุบัน |
เบริลเลียมและแมกนีเซียมอยู่ที่ส่วนบนของหมู่ที่ 2
ท่อ ธาตุเบริลเลียม ตั้งอยู่ที่ส่วนบนของกลุ่มธาตุ และชี้ให้เห็นแล้วว่าหมู่ที่ 2 ไม่สม่ำเสมออย่างสมบูรณ์แบบ มักพบเชื่อมโยงกับแร่เบอริล ซึ่งเป็นแร่ในตระกูลเดียวกันกับอีเมอรัลด์และอะควาแมรีน เบริลเลียมโดดเด่นเพราะมีน้ำหนักเบาและแข็งแกร่งผิดปกติ จึงมีประโยชน์ในการผลิตชิ้นส่วนประสิทธิภาพสูงที่ต้องการมวลต่ำ อย่างไรก็ตาม เบริลเลียมเป็นวัสดุที่ต้องใช้ความระมัดระวังเป็นพิเศษในการทำงานในสภาพแวดล้อมอุตสาหกรรม เนื่องจากฝุ่นละเอียดอาจเป็นอันตรายหากสูดดมเข้าไป ดังนั้นจึงมีการจดจำเบริลเลียมทั้งในแง่สมรรถนะและการปฏิบัติด้วยความระมัดระวัง
แมกนีเซียมรู้สึกคุ้นเคยมากกว่า สัญลักษณ์เคมีของแมกนีเซียม คือแมกนีเซียม (Mg) ซึ่งเป็นหนึ่งในโลหะที่รู้จักกันดีที่สุดในกลุ่มนี้ เนื่องจากพบได้ในน้ำทะเล แร่ธาตุทั่วไป และระบบมีชีวิต มันเป็นโลหะที่มีน้ำหนักเบาอย่างยิ่ง และเมื่อเผาไหม้จะให้แสงสีขาวเข้มมาก นี่คือเหตุผลที่แมกนีเซียมถูกเชื่อมโยงมาโดยตลอดกับลูกไฟและวัสดุที่ลุกไหม้อย่างรุนแรง อย่างไรก็ตาม ในชีวิตประจำวัน ผู้คนส่วนใหญ่จะพบมันในรูปแบบที่อ่อนโยนกว่านั้น เช่น บทบาททางโภชนาการ สารลดกรดในกระเพาะ หรือโลหะผสมน้ำหนักเบาที่ใช้ในกรณีที่การลดมวลมีความสำคัญ
แคลเซียมและสตรอนเทียมในวัสดุทั่วไป
แคลเซียมเป็นสมาชิกกลุ่มที่ 2 ที่ผู้อ่านส่วนใหญ่รู้จักดีที่สุด มันปรากฏอยู่ในหินปูน ชอล์ก เปลือกหอย และกระดูก จึงเชื่อมโยงศาสตร์เคมีเข้ากับธรณีวิทยาและชีววิทยาได้ทันที สารประกอบคาร์บอเนตของแคลเซียม (calcium carbonate) คือสารที่คุ้นเคยในบริบทนี้ มันช่วยอธิบายว่าทำไมธาตุในครอบครัวเดียวกันจึงมีบทบาทสำคัญทั้งในการก่อตัวของถ้ำ หินก่อสร้าง และโครงร่างของสิ่งมีชีวิต ตัวโลหะแคลเซียมเองมีปฏิกิริยาสูง แต่สารประกอบของแคลเซียมนั้นมีอยู่ทั่วไป จึงทำให้ธาตุนี้มักรู้สึกคุ้นเคยมากกว่าที่จะรู้สึกแปลกประหลาด
สตรอนเทียมจะจำได้ง่ายขึ้นเมื่อเชื่อมโยงกับสี สัญลักษณ์ของสตรอนเทียม คือ Sr และ สโตรนเทียม พบได้เป็นหลักในแร่ซีเลสไทรต์ (celestite) และแร่สตรอนเทียนไอท์ (strontianite) ราชสมาคมเคมี (The Royal Society of Chemistry) บรรยายสตรอนเทียมว่าเป็นโลหะสีเงินอ่อนที่นุ่ม ลุกไหม้ในอากาศ และทำปฏิกิริยากับน้ำ เกลือของสตรอนเทียมมีชื่อเสียงในการให้สีแดงสดใสในพลุและสัญญาณไฟฉุกเฉิน แหล่งข้อมูลเดียวกันยังระบุถึงการใช้งานอื่นๆ เช่น วัสดุเรืองแสงในที่มืด และสตรอนเทียมคลอไรด์เฮกซาไฮเดรต (strontium chloride hexahydrate) ที่ใช้ในยาสีฟันสำหรับผู้ที่มีฟันไวต่อความร้อน-เย็น ซึ่งทำให้สตรอนเทียมเป็นตัวอย่างที่ดีของธาตุที่มีปฏิกิริยาทางเคมีสูง แต่โดยทั่วไปแล้วเราพบมันผ่านสารประกอบต่างๆ เป็นหลัก
บาริอุมและเรเดียมในบริบทขั้นสูงหรือเฉพาะทาง
ท่อ ธาตุบาริอุม มักถูกจดจำผ่านความหนักของมัน โดยทั่วไปแล้วจะเชื่อมโยงกับแร่บาริท (barite) และหนึ่งในสารประกอบที่คุ้นเคยที่สุดของมันคือบาริอัมซัลเฟต สารประกอบนี้มีความสำคัญเนื่องจากมีความละลายน้อยมาก ซึ่งช่วยอธิบายได้ว่าเหตุใดบาริอัมจึงสามารถปรากฏในสถานการณ์จริง เช่น ของเหลวสำหรับการเจาะรู (drilling fluids) และการถ่ายภาพทางการแพทย์ ขณะที่สารประกอบบาริอัมที่ละลายน้ำได้อื่นๆ กลับได้รับการจัดการอย่างระมัดระวังยิ่งขึ้นเนื่องจากข้อกังวลเรื่องพิษ บาริอัมจึงเตือนผู้อ่านว่า รูปแบบที่มีประโยชน์ของธาตุหมู่ที่ 2 มักเป็นสารประกอบ ไม่ใช่โลหะที่มีประกายเงาเอง
เรเดียมตั้งอยู่ที่ส่วนล่างสุดของครอบครัวธาตุนี้ แต่มันไม่ได้กลมกลืนเข้ากับสิ่งแวดล้อมอย่างเงียบๆ บน ตารางธาตุเรเดียม มุมมอง: Ra หมายถึงจุดที่กัมมันตภาพรังสีกลายเป็นคุณลักษณะสำคัญที่สุด แรเดียมเกิดขึ้นตามธรรมชาติเพียงในปริมาณเล็กน้อย โดยมักพบควบคู่ไปกับแร่ยูเรเนียม ประวัติศาสตร์แสดงให้เห็นว่าแรเดียมมีชื่อเสียงจากสีเรืองแสงและงานทดลองทางการแพทย์ในยุคแรก ๆ ปัจจุบัน ความอันตรายของมันมาจากการกัมมันตภาพรังสีมากกว่าพฤติกรรมของโลหะทั่วไป จึงต้องจัดการภายใต้การควบคุมอย่างเข้มงวด ในแง่ง่าย ๆ แล้ว แรเดียมยังคงจัดอยู่ในหมู่ที่ 2 แต่จะถูกพูดถึงโดยคำนึงถึงความปลอดภัยด้านนิวเคลียร์ไม่แพ้ด้านเคมี
เมื่อนำธาตุทั้งหกตัวนี้มาวางเรียงเคียงกัน ครอบครัวนี้ก็จะไม่ดูเหมือนรายการชื่อที่เปลือยเปล่าอีกต่อไป ขนาด ความไวในการทำปฏิกิริยา สารประกอบที่พบได้ทั่วไป รวมทั้งวิธีที่แต่ละธาตุปรากฏตัวในชีวิตจริง ก็เปลี่ยนแปลงไปตามลำดับเมื่อเคลื่อนตัวลงมาตามกลุ่ม รูปแบบที่เปลี่ยนแปลงนี้เองที่ทำให้หมู่ที่ 2 มีประโยชน์อย่างยิ่ง เพราะลำดับจากเบริลเลียมไปจนถึงแรเดียมเริ่มเผยให้เห็นแนวโน้ม (trends) แทนที่จะเป็นเพียงข้อเท็จจริงกระจัดกระจาย (trivia)
สมบัติของโลหะดินเชิงเบสและแนวโน้มของหมู่ที่ 2
การเปลี่ยนแปลงลำดับจากเบริลเลียมไปยังเรเดียมนั้นคือสิ่งที่ทำให้หมู่ที่ 2 มีประโยชน์ แทนที่จะท่องจำข้อเท็จจริงที่แยกจากกันหกข้อ คุณสามารถติดตามรูปแบบจำนวนหนึ่งที่เกิดซ้ำตลอดแนวคอลัมน์นี้ได้ ซึ่งรูปแบบที่สำคัญที่สุด คุณสมบัติของโลหะแอลคาไลน์เอิร์ธ ล้วนเกิดขึ้นจากคุณลักษณะร่วมเพียงอย่างเดียว นั่นคือ อะตอมแต่ละตัวมีอิเล็กตรอนสองตัวอยู่ในชั้นนอกสุด ซึ่งมักจะสูญเสียออกไป
เมื่อคุณเข้าใจว่าขนาดของอะตอม การบดบังอิเล็กตรอน และพลังงานไอออไนเซชันเปลี่ยนแปลงอย่างไรเมื่อเคลื่อนลงมาตามหมู่ ครอบครัวนี้ก็จะทำนายพฤติกรรมได้ง่ายขึ้นมาก คุณสมบัติเหล่านี้ ลักษณะเฉพาะของโลหะแอลคาไลน์เอิร์ธ ไม่ใช่เพียงข้อเท็จจริงสำหรับการสอบเท่านั้น แต่ยังอธิบายเหตุผลที่สมาชิกบางตัวทำปฏิกิริยาได้เร็วกว่า ทำไมสารประกอบบางชนิดจึงละลายได้ดีกว่าชนิดอื่น และเหตุใดแนวโน้มบางประการจึงจำเป็นต้องใช้ถ้อยคำอธิบายอย่างระมัดระวัง แทนที่จะใช้ลูกศรแบบง่ายๆ
คุณสมบัติร่วมของโลหะแอลคาไลน์เอิร์ธ
สมาชิกส่วนใหญ่ของหมู่ที่ 2 เป็นโลหะสีเงินที่มักจะสร้างสารประกอบ M 2+ไอออนและสร้างสารประกอบที่มีลักษณะเป็นไอออนิกเป็นส่วนใหญ่ ธาตุเหล่านี้ทำหน้าที่เป็นตัวลด เนื่องจากสูญเสียอิเล็กตรอน เมื่อเปรียบเทียบกับโลหะกลุ่ม 1 แล้ว ธาตุเหล่านี้โดยทั่วไปมีปฏิกิริยาน้อยกว่า แต่ก็ยังมีความกระตือรือร้นทางเคมีเพียงพอที่จะก่อตัวเป็นออกไซด์ คลอไรด์ คาร์บอเนต และซัลเฟตที่พบได้ทั่วไปหลายชนิด
วิธีง่ายๆ ในการจัดระเบียบ สมบัติทางเคมีของโลหะแอลคาไลน์เอิร์ธ คือการแยกสิ่งที่คงที่ออกจากสิ่งที่เปลี่ยนแปลง สิ่งที่คงที่คือสถานะออกซิเดชันปกติที่ +2 ส่วนสิ่งที่เปลี่ยนแปลงคือความง่ายในการที่แต่ละธาตุจะสูญเสียอิเล็กตรอนทั้งสองตัวนั้น นั่นคือจุดเริ่มต้นของแนวโน้มต่างๆ
แนวโน้มลงตามกลุ่ม 2 และความหมายของมัน
ข้อมูลที่รวบรวมโดย LibreTexts และคำอธิบายแนวโน้มจาก Save My Exams แสดงรูปแบบโดยรวมที่เหมือนกัน รัศมีอะตอมเพิ่มขึ้นจาก 112 พิโคเมตรสำหรับ Be เป็น 253 พิโคเมตรสำหรับ Ba ในขณะที่พลังงานไอออไนเซชันลำดับแรกลดลงจาก 900 เป็น 503 กิโลจูล/โมล กล่าวอย่างง่ายคือ อิเล็กตรอนชั้นนอกอยู่ห่างจากนิวเคลียสมากขึ้น และถูกบดบังโดยเปลือกภายในที่มากขึ้น จึงทำให้ถูกกำจัดออกไปได้ง่ายขึ้น
| แนวโน้ม | ทิศทางลงตามกลุ่ม 2 | เหตุผลเชิงเคมี | สิ่งนี้หมายถึงอะไรในทางปฏิบัติ |
|---|---|---|---|
| รัศมีอะตอม | เพิ่มขึ้น | แต่ละธาตุมีเปลือกอิเล็กตรอนเพิ่มขึ้นหนึ่งชั้น และมีการบดบังมากขึ้น | อะตอมที่มีขนาดใหญ่กว่าจะยึดอิเล็กตรอนชั้นนอกได้แน่นน้อยลง |
| พลังงานไอออไนเซชันลำดับที่หนึ่งและลำดับที่สอง | ลดลงโดยรวม | อิเล็กตรอนชั้นนอกอยู่ห่างจากนิวเคลียสมากขึ้น จึงทำให้แรงดึงดูดอ่อนแอลง | การเกิดไอออน M 2+ง่ายขึ้น |
| ความไวต่อปฏิกิริยา | เพิ่มขึ้นโดยรวม | พลังงานไอออไนเซชันที่ต่ำลงทำให้สูญเสียอิเล็กตรอนได้ง่ายขึ้น | ธาตุหมู่หนักกว่ามีปฏิกิริยากับกรด ออกซิเจน และมักจะกับน้ำอย่างรุนแรงมากขึ้น |
| จุดละลาย | โดยทั่วไปลดลง แต่ไม่เป็นไปอย่างเรียบเนียน | ไอออนโลหะที่มีขนาดใหญ่ขึ้นทำให้พันธะโลหะอ่อนแอลง แม้ว่าโครงสร้างก็มีผลเช่นกัน | ใช้คำว่า "โดยทั่วไป" ที่นี่ เพราะ Mg และ Ca ไม่สอดคล้องกับแนวโน้มที่เรียบง่ายอย่างสมบูรณ์แบบ |
| ความหนาแน่น | ไม่สม่ำเสมอ | มวล ขนาดอะตอม และการจัดเรียงของอะตอมโลหะเปลี่ยนแปลงไปพร้อมกัน | คุณไม่สามารถพิจารณาความหนาแน่นเป็นแนวโน้มลดลงอย่างง่ายๆ ได้ |
| ความสามารถในการละลายของไฮดรอกไซด์ | เพิ่มขึ้น | สมดุลระหว่างพลังงานแลตทิซและพลังงานไฮเดรชันเปลี่ยนแปลงไปเมื่อเคลื่อนลงมาตามหมู่ | ไฮดรอกไซด์ของธาตุหมู่หนักกว่าให้สารละลายที่มีความเป็นด่างมากขึ้น |
| ความสามารถในการละลายของซัลเฟต | ลดลง | พลังงานการไฮเดรตลดลงเมื่อขนาดของแคทไอออนเพิ่มขึ้น | สารประกอบ เช่น BaSO 4จะกลายเป็นสารที่ละลายน้ำได้น้อยมาก |
ความหนาแน่นและพฤติกรรมการหลอมเหลวเป็นแนวโน้มสองประการที่นักเรียนมักทำให้เรียบง่ายเกินไป ความหนาแน่นไม่เปลี่ยนแปลงตามเส้นตรง เนื่องจากทั้งมวลและปริมาตรต่างก็เปลี่ยนแปลงไป และอะตอมของโลหะไม่จัดเรียงตัวในรูปแบบเดียวกันในทุกผลึก การหลอมเหลวก็เช่นกัน ซึ่งจำเป็นต้องพิจารณาอย่างระมัดระวัง โดยทั่วไปแล้วจุดหลอมเหลวมีแนวโน้มลดลง เนื่องจากไอออนที่มีขนาดใหญ่ขึ้นทำให้โครงสร้างตาข่ายโลหะอ่อนแอลง แต่มากีเซียม (Mg) มีจุดหลอมเหลวต่ำผิดปกติที่ 650 องศาเซลเซียส ในขณะที่แคลเซียม (Ca) เพิ่มขึ้นถึง 842 องศาเซลเซียส ก่อนที่ค่าจะลดลงอีกครั้ง ดังนั้นหนึ่งในลักษณะที่ปลอดภัยที่สุด ของโลหะกลุ่มอัลคาไลน์เอิร์ธ คือข้อนี้: รูปแบบโดยรวมนั้นมีอยู่จริง แต่รายละเอียดทางกายภาพไม่เรียบเนียนสมบูรณ์แบบ
ความสามารถในการละลายก็มีคำเตือนในลักษณะเดียวกัน ไม่มีกฎข้อเดียวที่สามารถอธิบายความสามารถในการละลายของเกลือกลุ่ม 2 ทั้งหมดได้ ไฮดรอกไซด์มีแนวโน้มละลายน้ำได้ดีขึ้นเมื่อเคลื่อนตัวลงในกลุ่ม ขณะที่ซัลเฟตมีแนวโน้มละลายน้ำได้น้อยลง หากมีผู้กล่าวว่า "ความสามารถในการละลายเพิ่มขึ้นเมื่อเคลื่อนตัวลงในกลุ่ม 2" คำถามสำคัญคือ "สารประกอบใด?"
เหตุใดโลหะกลุ่มอัลคาไลน์เอิร์ธจึงทำปฏิกิริยาในแบบที่มันทำ
ดังนั้น, โลหะกลุ่มอัลคาไลน์เอิร์ธมีความไวต่อการเกิดปฏิกิริยาหรือไม่ ? ใช่ และคำตอบโดยทั่วไปคือ ความไวต่อการเกิดปฏิกิริยาจะเพิ่มขึ้นเมื่อเคลื่อนลงมาตามหมู่ สาเหตุก็คือเรื่องเดียวกันเกี่ยวกับอิเล็กตรอนที่กล่าวมาข้างต้น ค่าพลังงานไอออไนเซชันลำดับที่หนึ่งและสองที่ต่ำกว่า หมายความว่าอะตอมสามารถสูญเสียอิเล็กตรอนสองตัวได้ง่ายขึ้น และเข้าสู่สถานะ M 2+ได้เร็วขึ้น
สิ่งนี้ส่งผลต่อปฏิกิริยาจริง กล่าวคือ เมื่อเคลื่อนลงมาตามหมู่ ปฏิกิริยากับกรดเจือจางจะเร็วขึ้น ปฏิกิริยากับออกซิเจนจะรุนแรงขึ้น และธาตุที่มีมวลอะตอมมากกว่าจะถูกออกซิไดซ์ได้ง่ายขึ้น บันทึกของ Save My Exams ระบุว่า บาริอุมมีความไวต่อการเกิดปฏิกิริยาสูงพอที่ต้องเก็บไว้ใต้น้ำมัน ซึ่งเป็นหลักฐานเชิงปฏิบัติที่แสดงให้เห็นว่าแนวโน้มความไวต่อการเกิดปฏิกิริยาสามารถดำเนินไปได้ไกลแค่ไหน
- รัศมีอะตอมเพิ่มขึ้นเมื่อเคลื่อนลงมาตามหมู่ที่ 2
- พลังงานไอออไนเซชันลดลงเมื่อเคลื่อนลงมาตามหมู่ที่ 2
- ความไวต่อการเกิดปฏิกิริยาเพิ่มขึ้น เนื่องจากการสูญเสียอิเล็กตรอนสองตัวทำได้ง่ายขึ้น
- จุดหลอมเหลวและความหนาแน่นแสดงความผิดปกติ ดังนั้นจึงควรหลีกเลี่ยงกฎที่แน่นอนแบบสัมบูรณ์
- ไฮดรอกไซด์และซัลเฟตแสดงแนวโน้มความละลายที่ตรงข้ามกัน
รูปแบบเหล่านั้นทำให้กลุ่มธาตุในครอบครัวนี้สามารถทำนายพฤติกรรมได้ แต่ไม่สม่ำเสมออย่างสมบูรณ์แบบ ที่บริเวณด้านบนสุดของกลุ่ม แบริลเลียมเริ่มแสดงพฤติกรรมที่ผิดกฎไปแล้ว และแมกนีเซียมก็เพิ่มข้อยกเว้นอันเป็นที่พบเห็นได้ทั่วไปอีกประการหนึ่ง ซึ่งมีความสำคัญมากกว่าที่ผู้เริ่มต้นหลายคนคาดไว้
โลหะแอลคาไลกับโลหะแอลคาไลน์เอิร์ธ
แนวโน้มทั่วไปทำให้การเรียนรู้กลุ่มที่ 2 เป็นเรื่องง่ายขึ้น แต่กลุ่มธาตุนี้จะเริ่มขาดความหมายหากพิจารณาสมาชิกทุกตัวว่าเหมือนกันทั้งหมด คำเตือนที่ชัดเจนที่สุดคือ แบริลเลียม ขณะที่แมกนีเซียมก็เพิ่มข้อยกเว้นที่ใช้งานได้จริงในชีวิตประจำวันอีกประการหนึ่ง และเมื่อผู้คนเปรียบเทียบ โลหะแอลคาไลกับโลหะแอลคาไลน์เอิร์ธ ชื่อที่คล้ายคลึงกันอาจซ่อนความแตกต่างทางเคมีที่ลึกซึ้งมากไว้
เหตุใดแบริลเลียมจึงไม่แสดงพฤติกรรมเช่นเดียวกับโลหะกลุ่มที่ 2 ทั่วไป
BYJU'S อธิบายว่าแบริลเลียมเป็นธาตุที่ผิดปกติอย่างชัดเจนในกลุ่มที่ 2 เนื่องจากขนาดที่เล็กผิดปกติ พลังงานไอออไนเซชันสูง และความสามารถในการขั้วโพลาไรซ์ที่แข็งแรง ซึ่งส่งผลให้พฤติกรรมของมันไม่เป็นไปตามลักษณะทั่วไปของกลุ่มธาตุนี้ ในภาษาที่เข้าใจง่าย ธาตุ Be 2+ดึงดูดกลุ่มอิเล็กตรอนที่อยู่ใกล้เคียงอย่างรุนแรง ทำให้สารประกอบของเบริลเลียมมักมีลักษณะเป็นโควาเลนต์มากกว่าสารประกอบที่มีลักษณะไอออนิกมากกว่าซึ่งเกิดจากธาตุในหมู่เดียวกันที่มีมวลอะตอมมากกว่า แหล่งข้อมูลเดียวกันยังระบุว่า เบริลเลียมมีจุดหลอมเหลวและจุดเดือดสูงกว่าธาตุอื่นในหมู่เดียวกัน และไม่ทำปฏิกิริยากับน้ำเช่นเดียวกับธาตุเพื่อนร่วมหมู่
แมกนีเซียมไม่แปลกประหลาดเท่าเบริลเลียม แต่ก็อาจดูมีปฏิกิริยาน้อยกว่าที่นักเรียนคาดไว้ แหล่งข้อมูล LibreTexts ชี้ว่า แมกนีเซียมที่สะอาดมาก ๆ จะทำปฏิกิริยากับน้ำเย็นเพียงเล็กน้อยเท่านั้น และปฏิกิริยาจะค่อย ๆ ชะลอลงอย่างรวดเร็ว เนื่องจากแมกนีเซียมไฮดรอกไซด์ซึ่งแทบไม่ละลายน้ำเกิดขึ้นเป็นชั้นฟิล์มปิดผิวหน้า สำหรับเรเดียมซึ่งอยู่ที่ปลายล่างสุดของหมู่ ปกติจะถูกพูดถึงแยกต่างหาก เนื่องจากกัมมันตภาพรังสีของมันมีบทบาทโดดเด่นเหนือการใช้งานจริงและการอภิปรายด้านความปลอดภัย
ความแตกต่างระหว่างโลหะแอลคาไลน์เอิร์ธกับโลหะแอลคาไล
ในเชิงง่าย แอลคาไล เทียบกับ แอลคาไลน์เอิร์ธ กล่าวคือ โลหะกลุ่ม 1 สูญเสียอิเล็กตรอนชั้นนอกหนึ่งตัว ในขณะที่โลหะกลุ่ม 2 สูญเสียอิเล็กตรอนสองตัว ความแตกต่างเพียงข้อนี้เองที่กำหนดลักษณะสมบัติของโลหะแอลคาไลและโลหะแอลคาไลน์เอิร์ธ ลักษณะสมบัติของโลหะแอลคาไลและโลหะแอลคาไลน์เอิร์ธ มากกว่าปัจจัยอื่นใดเกือบทั้งหมด
| คุณลักษณะ | โลหะอัลคาไล หมู่ 1 | โลหะอัลคาไลน์เอิร์ธ หมู่ 2 |
|---|---|---|
| อิเล็กตรอนวาเลนซ์ | 1 | 2 |
| ไอออนทั่วไป | M + | M 2+ |
| ปฏิกิริยากับน้ำเย็น | มักเกิดอย่างรุนแรงหรือแม้แต่รุนแรงมาก ทำให้เกิดไฮดรอกไซด์และไฮโดรเจน | ไม่สม่ำเสมอ: เบอริลเลียม (Be) ไม่ทำปฏิกิริยากับน้ำ แมกนีเซียม (Mg) ทำปฏิกิริยาอย่างอ่อน แคลเซียม (Ca), สตรอนเทียม (Sr) และบาริอุม (Ba) ทำปฏิกิริยาด้วยความรุนแรงที่เพิ่มขึ้นตามลำดับ |
| เคมีของออกซิเจนที่พบได้ทั่วไป | สามารถสร้างออกไซด์ เพอร์ออกไซด์ หรือซูเปอร์ออกไซด์ได้ | มักสร้างโมโนออกไซด์เป็นส่วนใหญ่; ออกไซด์ส่วนใหญ่เหล่านี้ทำปฏิกิริยากับน้ำให้เกิดไฮดรอกไซด์ แต่เบอริลเลียมออกไซด์ (BeO) เป็นข้อยกเว้น |
ข้อยกเว้นสำคัญที่นักเรียนมักมองข้าม
- ไม่ใช่ทุกธาตุในหมู่ 2 ที่ทำปฏิกิริยากับน้ำในลักษณะเดียวกัน
- สารประกอบเบริลเลียมมีลักษณะเป็นโควาเลนต์มากกว่าธาตุอื่นในกลุ่มเดียวกัน
- อย่าสับสน โลหะแอลคาไลและโลหะแอลคาไลน์เอิร์ธ ว่าเป็นกลุ่มเดียวกันเพียงเพราะชื่อฟังดูเกี่ยวข้องกัน
- ท่อ สมบัติของโลหะแอลคาไลและโลหะแอลคาไลน์เอิร์ธ ควรเรียนรู้เป็นรูปแบบที่มีข้อยกเว้น ไม่ใช่เป็นคำขวัญที่ตายตัว
นั่นยังเป็นวิธีที่ดีที่สุดในการเข้าใจ สมบัติทางเคมีของโลหะแอลคาไลและโลหะแอลคาไลน์เอิร์ธ . รูปแบบการจัดเรียงอิเล็กตรอนให้กฎพื้นฐาน แต่สารจริงจะเพิ่มมิติความซับซ้อน และมิตินั้นจะชัดเจนยิ่งขึ้นเมื่อคุณพิจารณาตำแหน่งที่ธาตุในหมู่ที่ 2 เกิดขึ้นจริงในธรรมชาติ: พบได้ยากมากในฐานะโลหะบริสุทธิ์ แต่มักพบได้บ่อยกว่ามากภายในแร่ธาตุ หิน น้ำทะเล กระดูก และสารประกอบเชิงอุตสาหกรรม
การเกิดของโลหะแอลคาไลน์เอิร์ธในธรรมชาติ
หากคุณนึกภาพ โลหะอัลคาไลน์เอิร์ธ เป็นตัวอย่างที่มีความแวววาวและบริสุทธิ์วางอยู่ในหิน ธรรมชาติกลับทำงานแตกต่างออกไป ธาตุในหมู่ที่ 2 มีความไวต่อปฏิกิริยาเพียงพอที่มักปรากฏในรูปของไอออนภายในแร่ธาตุ เกลือ หิน น้ำทะเล กระดูก และเปลือกหอย มากกว่าที่จะปรากฏในรูปของโลหะแบบไม่รวมตัว ไม่ว่าผู้คนจะค้นหาด้วยคำว่า โลหะอัลคาไลน์เอิร์ธ หรือคำที่ใช้กันทั่วไปมากกว่า รูปแบบตามธรรมชาติก็ยังคงเหมือนเดิม: ธาตุในกลุ่มนี้มีแนวโน้มที่จะเกิดเป็นสารประกอบอย่างแข็งขัน
รูปแบบนั้นเกิดขึ้นโดยตรงจาก สมบัติทางเคมีของโลหะอัลคาไลน์เอิร์ธ 2+ 2+เมื่อเกิดเหตุการณ์นั้นแล้ว ไอออนออกซิเจน คาร์บอเนต ซัลเฟต และฮาไลด์จะจับกับธาตุเหล่านี้อย่างรวดเร็วเพื่อสร้างสารประกอบแข็งที่สามารถคงอยู่ได้ทั้งในด้านธรณีวิทยาและชีววิทยา
เหตุใดโลหะอัลคาไลน์เอิร์ธจึงไม่พบในธรรมชาติในรูปโลหะแบบไม่รวมตัว
บริตันนิกา และ ThoughtCo ทั้งสองแหล่งอธิบายกลุ่ม 2 ว่าเป็นธาตุที่มีปฏิกิริยาสูง ซึ่งอธิบายได้ว่าเหตุใดธาตุเหล่านี้จึงพบได้ยากในรูปแบบที่ไม่รวมตัวกับธาตุอื่น ในอากาศ ธาตุหลายชนิดในกลุ่มนี้เกิดการสร้างชั้นออกไซด์ขึ้นอย่างรวดเร็ว ในสภาพแวดล้อมตามธรรมชาติ ธาตุเหล่านี้จะถูกทำให้มีเสถียรภาพยิ่งขึ้นอีกผ่านการเกิดเป็นคาร์บอเนต ซัลเฟต ซิลิเกต ฟลูออไรด์ หรือคลอไรด์ นี่คือเหตุผลที่แคลเซียมปรากฏอยู่ในหินปูนและเปลือกหอย แมกนีเซียมพบได้ในแร่ธาตุและน้ำทะเล ส่วนสตรอนเทียมหรือแบเรียมพบได้ในแหล่งแร่ ส่วนเรเดียมนั้นมีความหายากยิ่งกว่า โดยเกิดขึ้นเพียงในปริมาณเล็กน้อยเท่านั้นในแร่ยูเรเนียม
แร่และสารประกอบทั่วไปของกลุ่ม 2
| ธาตุ | แหล่งธรรมชาติที่พบโดยทั่วไป | สารประกอบที่คุ้นเคย | เหตุใดสารประกอบนั้นจึงมีความสำคัญ |
|---|---|---|---|
| เบริลลียม | เบอริล | BeO | เบอริลเป็นแหล่งเชิงพาณิชย์ของธาตุนี้ ในขณะที่ออกไซด์ของเบอริลเลียมเป็นสารประกอบที่สำคัญในวัสดุพิเศษ |
| แมกนีเซียม | แมกนีไซต์ โดโลไมต์ น้ำทะเล | MgCO 3หรือ Mg(OH) 2 | แสดงให้เห็นว่าทำไมแมกนีเซียมจึงพบได้บ่อยกว่าในรูปของแร่ธาตุ น้ำทะเล และยา มากกว่าในรูปของโลหะบริสุทธิ์ |
| แคลเซียม | หินปูน ชอล์ก หินอ่อน ยิปซัม กระดูก เปลือกหอย | คาโก 3 | เชื่อมโยงธรณีวิทยา วัสดุก่อสร้าง และโครงร่างภายในร่างกายเข้าด้วยกันในสารประกอบที่พบได้ทั่วไปมากชนิดหนึ่ง |
| สโตรนเทียม | เซเลสไตต์ แสตรนเชียนไอต์ | SrSO 4หรือ SrCO 3 | แร่เหล่านี้เป็นแหล่งธรรมชาติหลักของสารประกอบสตรอนเทียม |
| บารีียม | แบไรต์ วิเทไรต์ | BaSO 4 | แบไรต์เป็นแร่สำคัญ และแบเรียมซัลเฟตเป็นหนึ่งในสารประกอบแบเรียมที่คุ้นเคยมากที่สุด |
| เรเดียม | มีอยู่ในปริมาณน้อยมากในไพธ์บลินด์และแร่ยูเรเนียมอื่นๆ | RaCl 2 | ความหายากและความเป็นกัมมันตรังสีของเรเดียมทำให้สารประกอบเรเดียมมีความสำคัญทางประวัติศาสตร์ แต่กลับพบได้ยาก |
EBSCO ระบุว่า แคลเซียมและแมกนีเซียมยังพบได้ในน้ำทะเลที่ความเข้มข้นประมาณ 0.4 กรัม/ลิตร และ 1.3 กรัม/ลิตร ตามลำดับ ซึ่งช่วยอธิบายเหตุผลที่ธาตุกลุ่มนี้ โลหะกลุ่มดินแข็ง-ด่าง ไม่เพียงเชื่อมโยงกับแร่ธาตุเท่านั้น แต่ยังเชื่อมโยงกับน้ำแข็ง (hard water), ระบบนิเวศทางทะเล และเนื้อเยื่อของสิ่งมีชีวิตด้วย
วิธีการแยกโลหะเหล่านี้ออกจากสารประกอบของมัน
เนื่องจากโลหะกลุ่มที่ 2 มักถูกผูกมัดอยู่ภายในสารประกอบ กระบวนการสกัดจึงเริ่มต้นจากแร่ น้ำเกลือ (brines) หรือแหล่งสะสมแร่เป็นหลัก แนวคิดอุตสาหกรรมทั่วไปค่อนข้างเรียบง่าย คือ ขั้นตอนแรกแปลงวัสดุให้กลายเป็นออกไซด์หรือฮาไลด์ที่สามารถจัดการได้ง่ายกว่า จากนั้นจึงใช้กระบวนการอิเล็กโทรไลซิสหรือการลดด้วยสารเคมีเพื่อปลดปล่อยโลหะออกมา หนังสือสารานุกรมบริแทนนิกา (Britannica) กล่าวถึงการแยกแมกนีเซียม แคลเซียม สตรอนเทียม และแบเรียมครั้งแรกโดยวิธีอิเล็กโทรไลซิส ในขณะที่แหล่งข้อมูล EBSCO ชี้ว่า การผลิตในปัจจุบันยังคงพึ่งพาการใช้คลอไรด์หลอมเหลว การลดออกไซด์ หรือเส้นทางที่เกี่ยวข้องเป็นหลัก ขึ้นอยู่กับธาตุแต่ละชนิด เบอริลเลียมเป็นตัวอย่างที่เตือนใจว่า กลุ่มธาตุนี้ไม่สม่ำเสมออย่างสมบูรณ์แบบ เพราะเบอริลเลียมสามารถผลิตได้โดยการลดเบอริลเลียมฟลูออไรด์
ดังนั้น ในชีวิตประจำวัน ผู้คนมักจะพบธาตุกลุ่มที่ 2 ผ่านหินปูน ปูนปลาสเตอร์ แมกนีเซียมจากน้ำทะเล บาริเทต หรือแคลเซียมจากสิ่งมีชีวิต ไม่ใช่ผ่านตัวอย่างโลหะบริสุทธิ์ รายละเอียดข้อนี้มีความสำคัญ เพราะความสำคัญในโลกแห่งความเป็นจริงของธาตุเหล่านี้ขึ้นอยู่กับสารประกอบและรูปแบบของมันมากกว่าโลหะบริสุทธิ์เอง
ตัวอย่างของโลหะอัลคาไลน์เอิร์ธในชีวิตประจำวัน
กลุ่มที่ 2 จะจดจำได้ง่ายขึ้นมากเมื่อคุณเชื่อมโยงแต่ละธาตุเข้ากับสิ่งของหรือปรากฏการณ์ที่เกิดขึ้นจริง เช่น กระดูก ยาลดกรด ปูนปลาสเตอร์ ดอกไม้ไฟ ของเหลวสำหรับการเจาะน้ำมัน และหน้าปัดเรืองแสงแบบเก่า ล้วนเป็นตัวอย่างที่มีประโยชน์ โลหะอัลคาไลน์เอิร์ธ หากคุณเคยสงสัย แมกนีเซียมเป็นโลหะหรือไม่ใช่โลหะ หรือ ca เป็นโลหะหรือไม่ คำตอบทั้งสองข้อนั้นเรียบง่ายมาก: แมกนีเซียมและแคลเซียมเป็นโลหะ อย่างไรก็ตาม ในชีวิตประจำวัน ผู้คนมักจะพบธาตุเหล่านี้ในรูปของสารประกอบ ไม่ใช่ในรูปของตัวอย่างโลหะบริสุทธิ์
การใช้งานในชีวิตประจำวันของสารประกอบแมกนีเซียมและแคลเซียม
- แมกนีเซียม : แมกนีเซียมเป็นหนึ่งในธาตุที่มีความสำคัญทางชีวภาพมากที่สุด ธาตุกลุ่มอัลคาไลน์เอิร์ธ . แผ่นข้อมูลข้อเท็จจริงเกี่ยวกับแมกนีเซียมจากสถาบันสุขภาพแห่งชาติ (NIH) ระบุว่าแมกนีเซียมทำหน้าที่เป็นโคแฟคเตอร์ในระบบเอนไซม์มากกว่า 300 ระบบ และช่วยสนับสนุนการทำงานของกล้ามเนื้อและระบบประสาท การผลิตพลังงาน และโครงสร้างของกระดูก สารประกอบแมกนีเซียมยังปรากฏอยู่ในยาลดกรดและยาระบายบางชนิด ส่วนโลหะแมกนีเซียมมีคุณค่าในโลหะผสมน้ำหนักเบา ซึ่งการลดมวลมีความสำคัญ
- แคลเซียม สารประกอบแคลเซียมมีบทบาทเด่นในชีวิตประจำวัน แคลเซียมช่วยให้โครงสร้างของกระดูกและฟันแข็งแรง และสารประกอบ เช่น แคลเซียมคาร์บอเนตและแคลเซียมซัลเฟต มีบทบาทสำคัญในหินปูน ปูนซีเมนต์ ปูนปลาสเตอร์ และแผ่นยิปซัม ซึ่งทำให้แคลเซียมเป็นหนึ่งในธาตุที่เชื่อมโยงอย่างชัดเจนระหว่างเคมี ชีววิทยา และการก่อสร้าง
การใช้งานเฉพาะทางของสตรอนเทียมและแบเรียม
- สโตรนเทียม เกลือสตรอนเทียมเป็นที่รู้จักกันดีที่สุดในการให้สีแดงเข้มในดอกไม้ไฟและสัญญาณค้นหา แม้แต่ผู้อ่านที่อาจไม่จำรายชื่อธาตุกลุ่มที่ 2 ได้ครบถ้วน ก็มักจะจำสตรอนเทียมได้เมื่อเชื่อมโยงกับสี
- บารีียม สารประกอบแบเรียมมีความสำคัญทั้งในอุตสาหกรรมและการแพทย์ โปรไฟล์สารประกอบแบเรียมจากห้องสมุดแพทย์แห่งชาติ (NLM) อธิบายการใช้งานหลักในโคลนเจาะ สารเคลือบ พลาสติก อิฐ และแก้ว นอกจากนี้ยังระบุถึงการใช้งานด้านการแพทย์ที่สำคัญคือ บาริอัมซัลเฟตที่มีความละลายน้อยมาก ซึ่งใช้เป็นสารที่ขัดแสงรังสีเอ็กซ์ (radiopaque material) ในการตรวจเอกซเรย์บางประเภท เนื่องจากโดยทั่วไปแล้วร่างกายไม่ดูดซึมสารนี้
- เรเดียม : เรเดียมส่วนใหญ่มีการใช้งานในเชิงประวัติศาสตร์ หรืออยู่ภายใต้การควบคุมอย่างเข้มงวดในงานวิจัยทางวิทยาศาสตร์ หน้าเว็บไซต์ของ NRC เกี่ยวกับเรเดียม อธิบายการใช้งานในอดีตของเรเดียมในสีเรืองแสงและการรักษาโรคมะเร็งในช่วงแรก ปัจจุบันการใช้งานส่วนใหญ่เหล่านี้ถูกแทนที่แล้ว แม้ว่าจะยังคงมีการใช้งานที่ได้รับการควบคุมอยู่บ้าง เช่น การถ่ายภาพรังสีอุตสาหกรรม (industrial radiography) บางประเภท
เหตุใดรูปแบบและชนิดของสารประกอบจึงมีความสำคัญต่อการใช้งานจริง
สำหรับธาตุกลุ่ม 2 รูปแบบที่ผู้คนมักใช้งานจริงคือสารประกอบ ไม่ใช่ธาตุบริสุทธิ์
แนวคิดเพียงข้อเดียวนี้ช่วยคลี่คลายความสับสนได้มาก แมกนีเซียมที่อยู่ในอาหารหรือยาไม่ใช่สิ่งเดียวกับแมกนีเซียมริบบอนที่กำลังลุกไหม้ แคลเซียมในกระดูกไม่ใช่สิ่งเดียวกับโลหะแคลเซียมที่มีปฏิกิริยาแรง แบเรียมเป็นตัวอย่างที่ชัดเจนที่สุดว่าทำไมรูปแบบของธาตุจึงมีความสำคัญ: แบเรียมซัลเฟตที่ไม่ละลายน้ำสามารถนำไปใช้ประโยชน์ได้ในการถ่ายภาพทางการแพทย์ ขณะที่สารประกอบแบเรียมที่ละลายน้ำได้มากกว่านั้นจำเป็นต้องระมัดระวังอย่างยิ่ง ราเดียมยิ่งเน้นประเด็นนี้ให้ชัดเจนยิ่งขึ้น เพราะการจัดการกับมันขึ้นอยู่กับกัมมันตภาพรังสีของมัน ไม่ใช่เพียงแค่ตำแหน่งของมันในหมู่ธาตุโลหะเท่านั้น
ดังนั้น คุณค่าของหมู่ที่ 2 จึงไม่ใช่สิ่งที่นามธรรมแต่อย่างใด ธาตุเหล่านี้ช่วยอธิบายว่าทำไมธาตุในครอบครัวเดียวกันจึงมีบทบาทสำคัญทั้งในด้านโภชนาการ วัสดุศาสตร์ การแพทย์ การแปรรูปอุตสาหกรรม และกฎระเบียบด้านความปลอดภัย รายชื่อการใช้งานจริงที่สั้นๆ มักจะเพียงพอที่จะทำให้รูปแบบโดยรวมนั้นจดจำได้อย่างมั่นคง
ประเด็นสำคัญเกี่ยวกับธาตุหมู่ที่ 2
เมื่อถึงจุดนี้ คำว่า ธาตุหมู่ดินเชิงเบส ควรรู้สึกเหมือนไม่ใช่รายการที่ต้องท่องจำอีกต่อไป แต่กลับรู้สึกเหมือนรูปแบบหนึ่งที่คุณสามารถอ่านออกได้โดยตรงจาก คอลัมน์หมู่ที่ 2 บนตารางธาตุ หากยังมีผู้ใดถามว่า ธาตุอัลคาไลน์เอิร์ธคืออะไร คำตอบย่อสั้นๆ ยังคงเรียบง่าย: เบริลเลียม แมกนีเซียม แคลเซียม สตรอนเทียม แบเรียม และเรเดียม คำนิยามที่ครบถ้วนยิ่งขึ้นของ ธาตุอัลคาไลน์เอิร์ธ มีประโยชน์ยิ่งกว่า: ธาตุโลหะหกชนิดในหมู่ที่ 2 ซึ่งโดยทั่วไปจะสูญเสียอิเล็กตรอนชั้นนอกสองตัวและสร้างไอออน M 2+²⁺
ประเด็นสำคัญเกี่ยวกับธาตุอัลคาไลน์เอิร์ธ
- ตำแหน่งมีความสำคัญ: ธาตุทั้งหกนี้ ในหมู่ที่ 2 อยู่ในคอลัมน์ที่สองจากด้านซ้าย คือส่วนหมู่ที่ 2 ของบล็อก s
- สมาชิกในตระกูลมีจำนวนคงที่: เบริลเลียม (Be), แมกนีเซียม (Mg), แคลเซียม (Ca), สตรอนเชียม (Sr), บาริอุม (Ba) และเรเดียม (Ra) ประกอบขึ้นเป็นชุดทั้งหมด
- เคมีที่ร่วมกันอธิบายความคล้ายคลึงกันภายในตระกูล: อิเล็กตรอนชั้นนอกสุดของธาตุเหล่านี้อยู่ในวงโคจร ns 2รูปแบบการให้ค่าเวเลนซ์ทำให้เกิดไอออนประจุ +2 เป็นผลลัพธ์ที่พบได้ทั่วไป ซึ่งเป็นประเด็นหลักที่สรุปไว้โดย LibreTexts
- แนวโน้มหลักเมื่อเคลื่อนลงตามกลุ่มสามารถทำนายได้: รัศมีอะตอมเพิ่มขึ้น พลังงานไอออไนเซชันโดยทั่วไปลดลง และปฏิกิริยาเคมีมักเพิ่มขึ้นเมื่อเคลื่อนลงตามกลุ่ม
- ข้อยกเว้นมีความสำคัญ: เบริลเลียมแสดงพฤติกรรมเชิงโควาเลนต์มากกว่าธาตุอื่น ๆ แมกนีเซียมอาจดูมีปฏิกิริยาน้อยกว่าเนื่องจากชั้นผิวที่หุ้มอยู่ และเรเดียมมักถูกกล่าวถึงผ่านคุณสมบัติทางกัมมันตรังสีเป็นหลัก
- ในชีวิตจริง มักหมายถึงสารประกอบ ไม่ใช่โลหะบริสุทธิ์: ผู้คนมักพบกับแคลเซียมคาร์บอเนต แมกนีเซียมออกไซด์ และแบเรียมซัลเฟตบ่อยกว่าธาตุบริสุทธิ์ Ca, Mg หรือ Ba
ท่อ ตารางธาตุหมู่โลหะอัลคาไลน์เอิร์ธ คอลัมน์นี้จดจำได้ง่ายที่สุดในฐานะโลหะหกชนิดที่เชื่อมโยงกันด้วยกฎข้อเดียว: ธาตุเหล่านี้มักจะกลายเป็นไอออนที่มีประจุ +2 แต่แต่ละตัวแสดงกฎข้อนี้ในแบบที่แตกต่างกันเล็กน้อย
จากเคมีของหมู่ที่ 2 สู่ชิ้นส่วนโลหะที่ออกแบบขึ้น
ความรู้ด้านเคมีนี้ขยายออกไปไกลเกินหนังสือเรียน แหล่งข้อมูล LibreTexts ระบุว่า แมกนีเซียมในรูปธาตุผลิตในปริมาณมากและนำไปใช้ในโลหะผสมน้ำหนักเบาสำหรับโครงถังเครื่องบินและชิ้นส่วนเครื่องยนต์รถยนต์ คู่มือการใช้โลหะผสมฉบับกว้างขึ้นแสดงให้เห็นว่าเหตุใดสิ่งนี้จึงมีความสำคัญ: วิศวกรปรับองค์ประกอบและกระบวนการผลิตเพื่อสมดุลระหว่างน้ำหนัก ความแข็งแรง ความต้านทานการกัดกร่อน และความสามารถในการกลึงในชิ้นส่วนจริง
สำหรับผู้อ่านที่ย้ายจาก คอลัมน์หมู่ที่ 2 บนตารางธาตุ มุมมองไปสู่การผลิต เทคโนโลยีโลหะเส้าอี้ ให้ตัวอย่างที่เป็นรูปธรรมเกี่ยวกับความเชื่อมโยงนั้น หน้าเว็บเกี่ยวกับวัสดุสำหรับยานยนต์และการกลึงของบริษัทอธิบายกระบวนการผลิตชิ้นส่วนโลหะ ตั้งแต่ขั้นตอนการสร้างต้นแบบไปจนถึงการผลิตจำนวนมาก ซึ่งพฤติกรรมของวัสดุและการควบคุมกระบวนการต้องทำงานร่วมกันอย่างสอดคล้องกัน ตารางธาตุหมู่โลหะอัลคาไลน์เอิร์ธ จึงไม่ใช่เพียงแค่แผนภูมิสำหรับใช้ในห้องเรียนเท่านั้น แต่ยังเป็นส่วนหนึ่งของเหตุผลเชิงตรรกะในการเลือกโลหะและโลหะผสมสำหรับชิ้นส่วนวิศวกรรมที่ต้องมีน้ำหนักเบา ทนทาน และสามารถผลิตได้จริงอีกด้วย
คำถามที่พบบ่อยเกี่ยวกับธาตุหมู่เอิร์ธแอลคาไลน์
1. ธาตุหมู่เอิร์ธแอลคาไลน์มีทั้งหมดกี่ชนิด?
ธาตุหมู่เอิร์ธแอลคาไลน์มีทั้งหมด 6 ชนิด ได้แก่ เบอริลเลียม แมกนีเซียม แคลเซียม สตรอนเทียม แบเรียม และเรเดียม ธาตุเหล่านี้อยู่ในหมู่ที่ 2 ของตารางธาตุ และจัดอยู่ด้วยกันเนื่องจากโดยทั่วไปจะสูญเสียอิเล็กตรอนชั้นนอกสองตัว ทำให้เกิดรูปแบบไอออนที่มีประจุ +2 ร่วมกันในสารประกอบหลายชนิด
2. ธาตุหมู่เอิร์ธแอลคาไลน์แตกต่างจากธาตุหมู่แอลคาไลอย่างไร?
โลหะอัลคาไลอยู่ในหมู่ที่ 1 และมักจะสร้างไอออนที่มีประจุ +1 เนื่องจากมีอิเล็กตรอนชั้นนอกเพียงหนึ่งตัว ส่วนโลหะอัลคาไลน์เอิร์ธอยู่ในหมู่ที่ 2 มักจะสร้างไอออนที่มีประจุ +2 และโดยทั่วไปมีปฏิกิริยาน้อยกว่าโดยรวม อิเล็กตรอนวาเลนซ์ตัวที่เพิ่มขึ้นมานี้ส่งผลต่อความแข็งแรงของการเกิดพันธะ ปฏิกิริยากับน้ำ รวมถึงชนิดของเกลือและออกไซด์ที่พวกมันมักจะสร้างขึ้น
3. ทำไมโลหะอัลคาไลน์เอิร์ธจึงไม่พบในธรรมชาติในรูปแบบธาตุบริสุทธิ์?
โลหะเหล่านี้มีความไวต่อปฏิกิริยาพอสมควร จึงมักไม่คงอยู่ในรูปธาตุบริสุทธิ์เป็นเวลานานในสภาพแวดล้อมตามธรรมชาติ แต่จะทำปฏิกิริยากับไอออนออกซิเจน คาร์บอเนต ซัลเฟต คลอไรด์ หรือซิลิเกต แล้วกลายเป็นส่วนประกอบของแร่ธาตุ หิน น้ำทะเล เปลือกหอย และกระดูก นี่คือเหตุผลที่ผู้คนมักพบเจอธาตุในหมู่ที่ 2 ผ่านสารประกอบต่าง ๆ มากกว่าตัวอย่างโลหะบริสุทธิ์
4. โลหะอัลคาไลน์เอิร์ธทั้งหมดทำปฏิกิริยากับน้ำหรือไม่?
ไม่ นี่คือหนึ่งในข้อยกเว้นที่มีประโยชน์มากที่สุดที่ควรจดจำ แบริลเลียมมีความต้านทานต่อน้ำได้ดีมาก ในขณะที่แมกนีเซียมทำปฏิกิริยากับน้ำเย็นช้าลงเนื่องจากชั้นผิวที่เกิดขึ้นจำกัดการเกิดปฏิกิริยา ส่วนแคลเซียม สตรอนเทียม และบาริอุมจะทำปฏิกิริยากับน้ำได้ง่ายขึ้นโดยทั่วไปแล้ว ความสามารถในการทำปฏิกิริยากับน้ำจะเพิ่มขึ้นเมื่อเคลื่อนตัวลงมาตามหมู่ที่ 2
5. ทำไมโลหะกลุ่มดินแข็ง-ด่างจึงมีความสำคัญต่ออุตสาหกรรมและการผลิต?
ความสำคัญของโลหะเหล่านี้เกิดจากทั้งสารประกอบของพวกมันและบทบาทในการเลือกโลหะผสม แมกนีเซียมมีคุณค่าอย่างยิ่งในกรณีที่น้ำหนักเบาเป็นปัจจัยสำคัญ สารประกอบแคลเซียมมีบทบาทหลักในซีเมนต์และปูนปลาสเตอร์ ขณะที่สารประกอบบาริอุมถูกเลือกใช้สำหรับการประยุกต์ใช้งานเฉพาะทางในอุตสาหกรรมและทางการแพทย์ ในการผลิตจริง การเข้าใจพฤติกรรมของโลหะช่วยกำหนดแนวทางการกลึง การควบคุมเสถียรภาพของกระบวนการ และคุณภาพของชิ้นส่วน ซึ่งเป็นเหตุผลที่ผู้จัดจำหน่าย เช่น Shaoyi Metal Technology ให้ความสำคัญกับการกลึงชิ้นส่วนยานยนต์ที่ผ่านการรับรอง การควบคุมกระบวนการ และการสนับสนุนตั้งแต่ชิ้นส่วนต้นแบบจนถึงการผลิตจำนวนมาก
 ผลิตจำนวนน้อย แต่มีมาตรฐานสูง บริการสร้างต้นแบบอย่างรวดเร็วของเรามาพร้อมกับการตรวจสอบที่เร็วขึ้นและง่ายขึ้น —
ผลิตจำนวนน้อย แต่มีมาตรฐานสูง บริการสร้างต้นแบบอย่างรวดเร็วของเรามาพร้อมกับการตรวจสอบที่เร็วขึ้นและง่ายขึ้น —
