Ce sunt metalele din tabelul periodic? Numărul pe care cele mai multe pagini îl omit
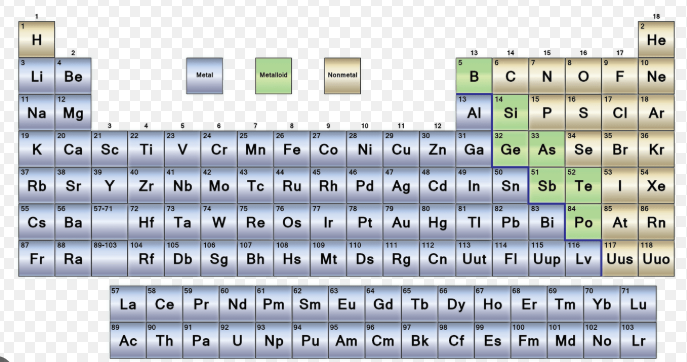
Care sunt metalele din tabelul periodic al elementelor?
Dacă ați căutat care sunt metalele din tabelul periodic al elementelor, răspunsul scurt este mai ușor decât pare la prima vedere. Metalele sunt elementele care se comportă, de obicei, în modul familiar metalic, cum ar fi conducerea electricității, reflexia luminii, deformarea fără rupere și pierderea electronilor în reacții.
Răspuns direct la întrebarea „Care sunt metalele din tabelul periodic?”
Metalele sunt elementele din tabelul periodic care prezintă, în general, comportament metalic. Majoritatea sunt buni conductori ai căldurii și ai electricității, au adesea luciu, sunt, de obicei, maleabile și ductile și tind să formeze ioni pozitivi prin pierderea electronilor. Majoritatea elementelor cunoscute sunt metale, deși numărul exact poate varia ușor în funcție de modul în care sunt clasificate elementele aflate la limită.
Pe scurt, cititorii care întreabă care sunt elementele metalice din tabelul periodic se referă la grupul mare care include exemple familiare precum sodiul, aluminiul, fierul, cuprul, argintul și aurul. În chimia de bază, tabela este adesea prezentată ca trei categorii largi: metale, nemetale și metaloizi.
De ce majoritatea elementelor sunt clasificate ca metale
Majoritatea elementelor se încadrează în categoria metalelor datorită comportamentului electronilor din stratul exterior. Metalele cedează, de obicei, electroni mai ușor decât nemetalele, ceea ce explică faptul că formează ioni pozitivi și că multe dintre ele conduc bine căldura și electricitatea. Britannica precizează că aproximativ trei pătrimi din elementele chimice cunoscute sunt metale, iar LibreTexts descrie metalele ca fiind elemente care formează, în mod obișnuit, ioni pozitivi prin pierderea electronilor.
- Majoritatea elementelor de pe tabel sunt metale.
- Caracteristicile cheie includ conductibilitatea, luciul, maleabilitatea și ductilitatea.
- Metalele cedează, de obicei, electroni în timpul reacțiilor chimice.
- Modelul metalelor și nemetalelor din tabelul periodic devine mai ușor de înțeles atunci când observați, de asemenea, grupul de delimitare al metaloizilor.
- Numărul exact de metale nu este întotdeauna prezentat în același mod pe fiecare diagramă.
Ultimul detaliu are o importanță mai mare decât pare la prima vedere, deoarece clasificarea începe cu proprietățile, dar aranjamentul tabelului periodic arată unde se găsesc, de obicei, metalele, nemetalele și metaloizii.
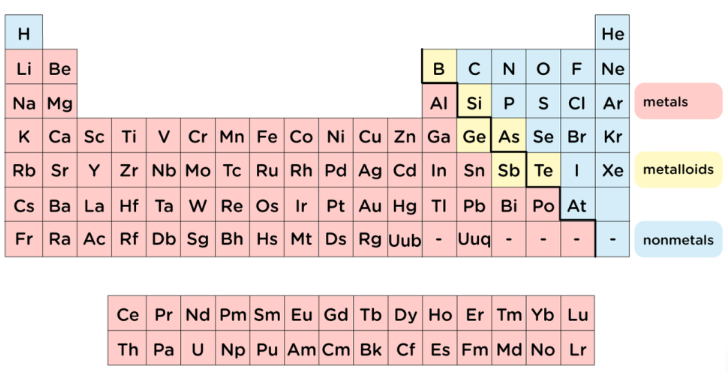
Unde sunt situate metalele în tabelul periodic?
O privire rapidă asupra unei diagrame codificate cromatic relevă modelul de bază. Dacă vă întrebați unde sunt situate metalele în tabelul periodic, uitați-vă spre partea stângă și spre centrul larg al tabelului. Sodiul se află foarte spre stânga , fierul ocupă centrul, iar metalele precum aluminiul și aurul arată că elementele metalice se întind pe o mare parte a tabelului. Chiar și cele două rânduri care sunt, de obicei, plasate sub corpul principal al tabelului — lantanidele și actinidele — sunt, de asemenea, metalice.
Unde sunt situate metalele în tabelul periodic
Elevii care întreabă unde se află metalele în tabelul periodic pot folosi linia în zigzag, sau linia în scară, ca ghid. Elementele situate în stânga acestei linii sunt, de obicei, metale. Elementele situate în dreapta sunt, în mare parte, nemetale. Elementele de-a lungul frontierei sunt metaloizii. Un rezumat al aranjamentului din ThoughtCo plasează majoritatea metalelor pe partea stângă a tabelului periodic, în timp ce ChemistryTalk descrie nemetalele ca fiind grupate în partea dreaptă, iar metaloizii – de-a lungul frontierei în zigzag.
Deci, unde se găsesc, în practică, metalele în tabelul periodic? În principal în stânga liniei în scară și în întreaga zonă centrală. Aceasta răspunde, de asemenea, la întrebarea unde se află metalele în tabelul periodic conform majorității manualelor. O excepție cunoscută este hidrogenul. Acesta apare în colțul superior stâng, dar este un nemetal.
| Regiunea tabelului | Clasificare tipică | Exemple |
|---|---|---|
| Partea stângă și cea centrală | În mare parte metale | Sodiu, aluminiu, fier, aur |
| Limită în zigzag | În principal metaloizi | Siliciu, arsen, telur |
| Colțul superior drept | În principal nemetale | Oxigen, azot, clor |
O tabelă periodică simplă, colorată codificat, face acest model mult mai ușor de reținut la prima vedere.
Cum se modifică caracterul metalic pe perioade și grupe
Poziția nu este aleatorie. Aceasta reflectă comportamentul electronilor. LibreTexts explică faptul că caracterul metalic crește, în general, pe măsură ce cobori într-o grupă și spre stânga pe o perioadă. Pe măsură ce cobori într-o grupă, atomii devin mai mari, iar energia de ionizare scade, astfel încât electronii din stratul exterior sunt mai ușor de eliminat. Pe o perioadă, de la stânga la dreapta, atomii rețin electronii mai strâns, deci comportamentul metalic scade.
Această tendință ajută la explicarea faptului că sodiul este mai metalic decât elementele situate mai spre dreapta în aceeași linie și de ce colțul din stânga-jos conține metalele cele mai reactive. Fierul, aluminiul și aurul sunt toate metale, dar pozițiile lor sugerează că nu toate metalele se comportă în același mod. Harta este clară. Numărul, totuși, devine mai dificil de stabilit, deoarece cazurile limită nu se încadrează în fiecare tabel în exact același mod.
Tabelul periodic: metale, nemetale, metaloizi
Acest model din stânga și centrul tabelului face metalele ușor de identificat, dar numărarea lor este mai puțin riguroasă decât sugerează multe pagini. Royal Society observă că peste două treimi dintre elemente sunt metale în condiții ambientale. Totuși, surse diferite nu oferă întotdeauna același număr exact, deoarece răspunsul depinde de modul în care sunt tratate elementele aflate la limită în tabelul elementelor: metale, nemetale și metaloizi.
De ce sursele nu sunt de acord cu privire la numărul de metale
Dezacordul provine, de obicei, din regulile de clasificare, nu din numărarea incorectă. Aceeași recenzie a Royal Society subliniază un detaliu important: tabelul periodic listează elemente, dar etichetele precum „metal” și „nemetal” descriu modul în care aceste elemente se comportă în starea lor elementală, în condiții obișnuite. În apropierea „scării”, acest comportament nu este întotdeauna clar delimitat. Recenzia evidențiază, de asemenea, faptul că unele părți ale blocului p, în special din jurul grupurilor 14 și 15, pot străbate granița dintre metale și nemetale. Așadar, deși un diagramă didactică a metalelor din tabelul periodic nemetale, metaloizi este utilă, ea simplifică o realitate mai complexă.
Dacă o pagină indică un număr exact al metalelor, fără a specifica regulile aplicate, ordinea estetică poate prevala asupra acurateței.
Cum modifică regulile de clasificare numărul total
Un număr total conservator pornește de la familiile clar metalice. Un număr total mai larg poate include, de asemenea, elementele metalice din blocul p, tratând cu mai multă prudență elementele adiacente „scării”. IUPAC menține tabelul periodic actualizat și observă că chiar și întrebările structurale, cum ar fi plasarea în Grupa 3, au fost obiect de dezbatere. Această dezbatere nu anulează imaginea de ansamblu, dar reamintește cititorilor că clasificarea științifică include atât convenția, cât și observația. În practică, cel mai mare problemă de clasificare este de obicei regiunea de frontieră, unde eticheta de metal, nemetal sau metaloid poate varia de la un tabel la altul.
| Categorie | Tratament tipic | De ce contează |
|---|---|---|
| Familii clar metalice | Sunt aproape întotdeauna numărate ca metale | Include principalele blocuri metalice și generează puține dezacorduri |
| Elemente metalice din blocul p | Sunt de obicei numărate ca metale | Rămân încă metalice, dar sunt mai aproape de limita în formă de scară |
| Regiune de frontieră | Pot fi etichetate ca metaloizi sau intermediare | Aici este unde comparațiile dintre metaloizi, metale și nemetale creează totaluri diferite |
Un răspuns util, așadar, nu este doar un număr. Este o privire pe familii asupra grupurilor care sunt întotdeauna incluse și asupra celor care se află suficient de aproape de limită pentru a genera confuzie.
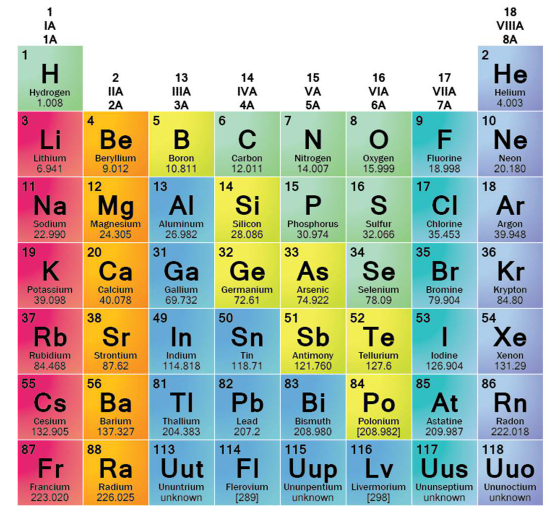
Familiile din Tabelul periodic al elementelor
O privire pe familii face partea metalică a tabelului mult mai ușor de înțeles. În chimie, o familie de elemente din tabelul periodic grupează elemente care au structuri similare ale electronilor din stratul exterior și, ca urmare, comportamente similare. De aceea, clasificarea în metale este mai utilă decât o simplă hartă stânga-versus-dreapta. O prezentare rapidă de la ThoughtCo, împreună cu clasificarea metalelor utilizată de Los Alamos , oferă cititorilor un mod practic de a sorta principalele familii metalice.
Familiile metalice din Tabelul periodic
Cele șase familii pe care le necesită majoritatea cititorilor sunt metalele alcaline, metalele alcalino-pământoase, metalele de tranziție, metalele post-tranziție, lantanidele și actinidele. Dacă ați văzut denumiri diferite pentru grupele din tabelul periodic, acest lucru este normal. Tabelele moderne numerotează coloanele de la 1 la 18, dar etichetele de familie se concentrează asupra trăsăturilor chimice comune, iar unele familii acoperă mai mult de o singură coloană sau chiar rândurile separate situate sub tabelul principal.
| Familia metalică | Unde apare | Trăsături de reținut |
|---|---|---|
| Metale alcaline | Grupa 1, cu excepția hidrogenului | Un electron de valență, moi, lucioase, foarte reactive, formează în mod obișnuit ioni +1 |
| Metale alcalino-pământoase | Grupul 2 | Doi electroni de valență, mai dure și mai dense decât metalele alcaline, formează în mod obișnuit ioni +2 |
| Metale de tranziție | Grupurile 3–12, blocul d central | Dure, dense, conductoare, adesea cu puncte de topire ridicate, mai multe stări de oxidare |
| Metale post-tranziționale | blocul p, la dreapta blocului de tranziție | Metale mai moi, care conduc mai slab decât metalele de tranziție |
| Lantanide | Elementele 57–71, prima linie separată | Proprietăți chimice foarte asemănătoare, parte a blocului f |
| Actinide | Elementele 89–103, a doua linie separată | metale din blocul f, toate radioactive |
Ce face diferit fiecare grup de metale
Începeți din partea cea mai stângă. Metalele alcaline din tabelul periodic sunt cele mai ușor de identificat, deoarece au un singur electron de valență și reacționează violent, în special cu apa. Metalele din Grupul 2 reacționează totuși, dar cei doi electroni exteriori le fac mai puțin reactive și, în general, mai dure decât cele din Grupul 1. În centrul tabelului periodic, blocul metalelor de tranziție include blocul central larg, cunoscut pentru solidele metalice dure, conductivitatea bună și gama largă de stări de oxidare.
Mutați-vă puțin mai spre dreapta și modelul se atenuează. Metalele post-tranziționale rămân metalice, dar sunt de obicei mai moi și conducători mai slabi decât metalele tranziționale. Cele două rânduri desenate sub tabel adaugă chiar mai multă nuanță. Lantanidele au o chimie strâns legată, în timp ce actinidele se remarcă prin radioactivitate. Unele surse descriu chiar ambele rânduri ca fiind metale tranziționale speciale, ceea ce arată de ce denumirile grupurilor din tabelul periodic pot ajuta, dar nu pot înlocui comportamentul chimic real.
- Grupul 1 înseamnă moale și foarte reactiv.
- Grupul 2 înseamnă reactiv, dar de obicei mai rezistent decât Grupul 1.
- Grupurile 3–12 înseamnă blocul central, care conține numeroase metale clasice.
- Post-tranziționale înseamnă metale mai moi, situate în regiunea „scării”.
- Lantanidele și actinidele înseamnă cele două rânduri ale blocului f, plasate sub corpul principal al tabelului.
Aceste etichete de familie fac tabelul mai organizat, dar testul mai profund al unui metal nu este doar numele său de familie. Conductivitatea, strălucirea, maleabilitatea și pierderea electronilor explică de ce toate aceste grupe se află, în primul rând, pe partea metalică.
Care sunt proprietățile metalelor?
Etichetele de familie facilitează parcurgerea tabelului periodic, dar chimistii identifică un metal după comportamentul său, nu doar după nume. Când elevii întreabă care sunt proprietățile metalelor, răspunsul începe cu un model de trăsături fizice și chimice comune. În LibreTexts descrierea legării metalice, atomii metalici sunt atrași de un „bazin” de electroni mobili și delocalizați. Acest model simplu ajută la explicarea proprietăților metalice ale metalelor și la înțelegerea motivului pentru care atât de multe familii diferite de metale împart, totuși, un set recunoscut de comportamente.
Proprietățile comune ale majorității metalelor
Dacă comparați proprietățile metalelor și ale nemetalelor, metalele se disting, de obicei, în câteva moduri clare.
- Conductivitate electrică: Electronii mobili permit metalelor să conducă bine curentul electric. Firul de cupru este exemplul clasic.
- Conductivitate termică: Acești aceiași electroni ajută la transferul căldurii, motiv pentru care metalele precum cuprul și aluminiul sunt utile acolo unde transferul de căldură este important.
- Luciu: LibreTexts explică faptul că electronii metalelor pot absorbi energie și apoi pot reemite lumină, conferind metalelor suprafața lor lucioasă. Aurul, argintul și cuprul ilustrează clar acest fenomen.
- Maleabilitate: Metalele pot fi lovite sau laminare în foi, în loc să se spargă. Folii de aluminiu și foi subțiri de aur sunt exemple ușor de înțeles.
- Ductilitate: Metalele pot fi trasate în fire. Cuprul este din nou un caz familiar.
- Formarea ionilor pozitivi: Multe metale pierd electroni în timpul reacțiilor. Sodiul formează Na+, magneziul formează Mg2+, iar aluminiul formează Al3+.
| Proprietate | Element reprezentativ | Ce arată |
|---|---|---|
| Conductivitate electrică | Cupru | Util pentru cabluri și circuite |
| Conductivitate termică | Aluminiu | Transferă eficient căldura |
| Stralucesc | Argint | Suprafață reflectorizantă, lucioasă |
| Maleabilitatea | Aur | Poate fi modelat în foi foarte subțiri |
| ELASTICITATE | Cupru | Poate fi tras în fire lungi |
Exemple care arată că metalele nu sunt toate la fel
Aceste trăsături sunt tendințe puternice, nu o listă de verificare perfectă. LibreTexts observă că mercurul este lichid la temperatura camerei, deși metalele sunt, de obicei, solide. Aceeași sursă subliniază faptul că sodiul și potasiul sunt suficient de moi pentru a fi tăiați cu un cuțit, ceea ce îi face foarte diferiți de un metal dur, cum ar fi fierul. Conductivitatea variază, de asemenea. Argintul și cuprul sunt conductori excepțional de buni, în timp ce unele metale au o performanță mai scăzută. Reactivitatea variază la fel de mult. Aurul își păstrează aspectul mai bine decât multe metale, deoarece rezistă coroziunii mult mai eficient decât metale precum fierul.
De aceea, caracteristicile metalelor sunt mai bine tratate ca un grup de indicii. Strălucirea singură nu este suficientă. Conductivitatea singură nu este suficientă. Chimistii analizează întregul model: modul în care un element conduce, se îndoaie și gestionează pierderea electronilor în reacții. Văzut astfel, următoarea întrebare practică devine mult mai ușor de răspuns: care elemente specifice aparțin categoriei metalelor atunci când le sortați familie cu familie?
Listă de metale după familie din tabelul periodic
Citiții care doresc o listă practică de metale nu au, de obicei, nevoie de un zid de denumiri ale elementelor. Ei au nevoie de structură. Gruparea elementelor metalice pe familii facilitează studierea, compararea și reținerea modelului. Tabelul principal de mai jos urmează clasificările generale ale metalelor utilizate de Science Notes și ThoughtCo, marcând în același timp cele câteva cazuri în care sursele de chimie le tratează uneori diferit. Aceasta este calea cea mai clară de a răspunde la întrebarea ce elemente sunt metale în tabelul periodic, fără a pretinde că fiecare etichetă marginală este universal fixată.
O listă pe familii a elementelor metalice
| Familia | Elemente din această familie | Notă privind clasificarea |
|---|---|---|
| Metale alcaline | Litiu, sodiu, potasiu, rubidiu, cesiu, franciu | Hidrogenul se află în Grupa 1, dar este de obicei tratat ca un nemetal în condiții obișnuite. |
| Metale alcalino-pământoase | Beriliu, magneziu, calciu, stronțiu, bariu, radiu | Acestea sunt întotdeauna clasificate ca metale. |
| Metale de tranziție | Scandiu, titan, vanadiu, crom, mangan, fier, cobalt, nichel, cupru, zinc, itriu, zirconiu, niobiu, molibden, tehneciu, ruteniu, rodium, paladiu, argint, cadmiu, hafniu, tantal, tungsten, reniu, osmiu, iridiu, platină, aur, mercur, rutherfordiu, dubniu, seaborgiu, bohriu, hassiu, meitneriu, darmstadtiu, roentgeniu, coperniciu | Majoritatea tabelelor didactice plasează Zn, Cd și Hg aici, deși unele discuții de chimie le tratează ușor diferit. |
| Metale post-tranziționale sau metale bazice | Aluminiu, Galiu, Indiu, Staniu, Talium, Plumb, Bismut, Poloniu, Nihonium, Flerovium, Moscovium, Livermorium | Notele de știință privind metalele de bază indică faptul că această grupă variază cel mai mult în funcție de sursă. Poloniul este adesea inclus, dar uneori este obiect de dezbatere. Livermorium este adesea tratat ca un metal posibil sau prevăzut. |
| Lantanide | Lantan, Ceriu, Praseodim, Neodim, Prometiu, Samariu, Europiu, Gadoliniu, Terbiu, Disprosiu, Holmiu, Erbiu, Tuliu, Iterbiu, Lutețiu | Acestea sunt primul rând separat, situat sub tabelul principal, și sunt metale. |
| Actinide | Actiniu, Toriu, Protactiniu, Uran, Neptuniu, Plutoniu, Americiu, Curium, Berkelium, Californiu, Einsteinium, Fermium, Mendeleviu, Nobelium, Lawrențiu | Acestea sunt al doilea rând separat, situat sub tabelul principal, și sunt metale, deși multe dintre ele sunt mai cunoscute pentru radioactivitate decât pentru comportamentul lor metalic obișnuit. |
Cum să citiți lista principală fără confuzie
Dacă aveți nevoie de o listă rapidă de metale pentru teme sau revizuire, folosiți mai întâi coloana familie și apoi coloana notă. Familia vă indică unde se află elementul în tabelul periodic. Nota vă arată unde clasificarea devine neclară. Acest lucru este cel mai important în apropierea scării și printre cele mai grele elemente din blocul p.
Când profesorii cer elevilor să enumere metalele , de obicei caută nucleul stabil al acestor familii, nu o dispută privind fiecare caz marginal. Dacă dorești doar numele celor mai familiare metale , începe cu membrii cei mai cunoscuți ai fiecărui grup și extinde lista treptat de la aceștia.
- Metale alcaline: sodiu, potasiu
- Metale alcalino-pământoase: magneziu, calciu
- Metale de tranziție: fier, cupru, argint, aur
- Metale post-tranziție: aluminiu, staniu, plumb
- Lantanide: lantan, neodim
- Actinide: uraniu, plutoniu
Acestea sunt unele exemple de metale pe care majoritatea cititorilor le recunosc deja. Ele constituie, de asemenea, repere memorabile utile atunci când întreaga tabelă pare prea încărcată. Pentru notele de studiu, este util să rețineți că metalele comune metale provind adesea din grupurile de tranziție și post-tranziție, în timp ce lantanidele și actinidele sunt mai ușor de reținut ca serii.
Un ultim avertisment menține această listă principală corectă: nu toate tabelele trasează aceeași limită în jurul elementelor precum poloniul sau cei mai grei membri sintetici ai blocului p. De aceea, o referință utilă face mai mult decât să denumească elementele. Arată, de asemenea, unde limitele devin neclare, deoarece o etichetă de „metal” este cea mai de încredere atunci când puteți distinge, de asemenea, un metal de un metaloid sau de un nemetal.
Ghid periodic al metalelor versus nemetalelor
O listă principală lungă este utilă, dar majoritatea cititorilor au nevoie de un mod mai rapid de a clasifica un element la prima vedere. Veste bună este faptul că tabela periodică vă oferă un indiciu vizual clar. Vesta și mai bună este aceea că chimia vă oferă un test suplimentar atunci când aranjamentul singur nu este suficient.
Cum se separă metalele de metaloizi și nemetale
O hartă vizuală din Science Notes evidențiază clar modelul de bază: metalele se află în principal în stânga și centrul tabelei, în timp ce nemetalele se grupează în dreapta. Între ele se află scara în zigzag cunoscută. Dacă vă întrebați unde sunt localizați metaloizii în tabela periodică, aceștia se găsesc, de obicei, de-a lungul acestei limite în zigzag. Ghidul de chimie UMD folosește același model pentru identificarea rapidă.
Totuși, întrebarea privind diferențierea dintre metale și nemetale pe tabelul periodic nu este rezolvată doar prin poziția elementelor. Metalele și nemetalele de pe tabelele periodice sunt mai bine separate și în funcție de comportamentul lor. Metalele conduc, de obicei, bine căldura și electricitatea și tind să piardă electroni pentru a forma ioni pozitivi. Nemetalii de pe tabelul periodic sunt mai predispuși să câștige sau să împartă electroni, iar mulți dintre ei sunt conductori slabi. Metaloizii de pe tabelul periodic se află între aceste două categorii, prezentând adesea proprietăți mixte și un comportament semiconductor.
- Găsiți linia în formă de scară pe tabel.
- Priviți mai întâi spre stânga sau spre centrul tabelului. Majoritatea elementelor de acolo sunt metale.
- Priviți în colțul din dreapta sus. Majoritatea elementelor de acolo sunt nemetale.
- Verificați chiar granița în sine. Elementele situate de-a lungul acesteia sunt adesea metaloizi.
- Verificați comportamentul, dacă este necesar. Un bun conductor indică un metal, un conductor slab indică un nemetal, iar un comportament intermediar sau semiconductor indică un metaloid.
- Observați excepțiile. Hidrogenul este plasat în stânga, dar este de obicei un nemetal. Dacă întrebați dacă siliciul este un metal, un nemetal sau un metaloid, siliciul este de obicei clasificat ca metaloid. Rolul său de semiconductor este evidențiat în ghidul MISUMI privind metaloizii.
Scara este un ghid, nu o garanție. Elementele din zona de graniță pot fi etichetate diferit, în funcție de tabel și de regulile de clasificare care stau la baza acestuia.
Ajutoare simple de memorare pentru identificare mai rapidă
- În stânga și în centru, gândiți-vă la metale.
- În partea de sus-dreapta, gândiți-vă la nemetale.
- Pe scara, gândiți-vă la metaloizi.
- Amintiți-vă indiciul comportamental: conduc, izolează sau semiconduc.
Acest cadru rapid facilitează citirea metalelor și nemetalelor pe diagramele tabelului periodic, chiar și în condiții de presiune. De asemenea, indică ceva mai important decât simpla memorare, deoarece diferența dintre un metal conductor și un metaloid semiconductor determină modul în care sunt alese materialele reale în domeniul electronicului și al fabricației.

De ce contează metalele din tabelul periodic în fabricație
Modelul în spirală nu ajută doar elevii să sorteze elementele. În domeniul proiectării și al producției, întrebarea «ce este metalul» se transformă rapid într-o decizie practică privind performanța. Cunoașterea poziției metalelor din tabelul periodic oferă inginerilor un prim indiciu despre conductivitate, rezistență, ductilitate și transferul de căldură, dar fabricarea reală merge mai departe decât etichetele din clasă.
De ce este importantă clasificarea metalelor în producția industrială reală
Un element chimic metalic este adesea punctul de plecare, nu cel de sosire. AJProTech descrie selecția materialelor ca pe un echilibru între sarcini, mediu, greutate, posibilități de fabricație, disponibilitate, cost și conformitate. De aceea, diferite tipuri de metale rezolvă probleme diferite. TIRapid evidențiază clar acest model: cuprul este apreciat pentru conductivitatea electrică și termică, aluminiul pentru densitatea scăzută și rezistența la coroziune, oțelul pentru rezistență și eficiență din punct de vedere al costurilor, iar titanul pentru rezistența specifică ridicată în medii solicitante. În practică, multe piese finite folosesc aliaje, nu elemente chimice metalice pure, deoarece sarcina necesită, de obicei, un echilibru mai bun al proprietăților.
- Transport: Aluminiul și magneziul contribuie la reducerea greutății, în timp ce oțelul rămâne o alegere frecventă pentru piesele structurale, deoarece combină rezistența cu un cost practic.
- Electronice: Cuprul este preferat acolo unde sunt esențiale trecerea curentului electric și transferul de căldură.
- Mediu greu: Oțelul inoxidabil, titanul și materialele pe bază de nichel sunt utile atunci când rezistența la coroziune sau stabilitatea la temperaturi ridicate devin critice.
- Planificarea producției: Prelucrarea prin așchiere are, de asemenea, importanță. Un material care pare ideal pe hârtie poate totuși duce la uzură crescută a sculelor, la prelungirea termenelor de livrare sau la creșterea cerințelor de inspecție.
Unde să explorați prelucrarea precisă a metalelor
Un element metalic din tabelul periodic devine un component util doar atunci când procesul de fabricație este potrivit pentru acel material. Aluminiul permite o prelucrare rapidă și proiectarea ușoară, în timp ce oțelurile mai rezistente sau aliajele de titan necesită un control mai strict al procesului. De aceea, inginerii iau în considerare nu doar compoziția chimică, ci și toleranțele, tratamentele de suprafață, validarea și reproductibilitatea.
Pentru un exemplu practic, Shaoyi Metal Technology prezintă un flux de lucru de prelucrare automotive care leagă prototiparea rapidă, producția de volum mic și producția de masă, cu sistemul de management al calității IATF 16949 și cu controlul statistic al proceselor. În acest mod de utilizare, tabelul periodic încetează să fie o schemă de memorat și devine un ghid pentru alegerea materialelor care pot fi prelucrate, inspectate și utilizate în componente reale, cu încredere.
- Folosiți compoziția chimică pentru a restrânge domeniul de opțiuni.
- Utilizați criterii de inginerie pentru a alege materialul final.
- Utilizați controlul procesului pentru a transforma metalul potrivit într-o piesă fiabilă.
Aceasta este valoarea reală pe care o aduce învățarea despre metalele din tabelul periodic: nu doar denumirea lor, ci și înțelegerea modului în care clasificarea metalelor influențează piesele cu care oamenii conduc, cablizează, răcesc și construiesc în fiecare zi.
Întrebări frecvente despre metalele din tabelul periodic
1. Câte metale sunt în tabelul periodic?
Nu există un singur număr pe care toate sursele să-l considere definitiv. Majoritatea elementelor sunt metale, dar numărul exact poate varia în funcție de modul în care o schemă tratează cazurile limită, în special în regiunea „scărilor” și printre unele elemente mai grele din blocul p. Un răspuns atent distinge clar familiile metalice netede de elementele care sunt uneori etichetate diferit, evitând astfel o cifră simplificată excesiv.
2. Unde se găsesc metalele în tabelul periodic?
Metalele se găsesc în principal pe partea stângă și în centrul tabelului periodic. Cele două rânduri separate de la partea de jos, lantanidele și actinidele, sunt, de asemenea, metale. Un mod rapid de a interpreta această dispunere este utilizarea liniei în formă de scară: majoritatea elementelor din stânga sunt metale, majoritatea celor din dreapta sunt nemetale, iar zona de tranziție conține numeroase metaloizi. Hidrogenul reprezintă excepția vizuală comună, deoarece este plasat în stânga, dar este de obicei clasificat ca nemetal.
3. Care sunt principalele familii de metale din tabelul periodic?
Principalele familii de metale sunt metalele alcaline, metalele alcalino-pământoase, metalele de tranziție, metalele post-tranziție, lantanidele și actinidele. Fiecare familie are propriul său model de comportament. Metalele alcaline sunt foarte reactive, metalele alcalino-pământoase sunt mai puțin reactive, dar totuși active, metalele de tranziție includ multe metale structurale și inginerești cunoscute, metalele post-tranziție sunt în general mai moi, iar lantanidele și actinidele formează cele două rânduri metalice afișate sub tabelul principal.
4. Ce proprietăți fac ca un element să fie un metal?
Chimiștii identifică de obicei un metal pe baza unui grup de trăsături, nu doar a unei singure caracteristici. Metalele conduc în mod obișnuit căldura și electricitatea foarte bine, reflectă lumina, se îndoaie fără a se rupe, se pot întinde în fire și tind să piardă electroni în reacții. Totuși, nu toate metalele se comportă în același fel. Unele sunt moi, altele rezistă foarte bine la coroziune, iar un exemplu bine-cunoscut, mercurul, este lichid la temperatura camerei.
5. De ce este important să știm dacă un element este un metal în domeniul fabricației?
Clasificarea metalelor ajută la stabilirea legăturii dintre chimie și alegerile reale de materiale. Odată ce inginerii știu că un material este metalic, pot începe să reflecteze asupra conductivității, rezistenței mecanice, rezistenței la coroziune, greutății și prelucrabilității acestuia. Aceste aspecte sunt esențiale în domeniul electronicului, al pieselor destinate transportului și al componentelor industriale. În practică, transformarea unui element metalic sau a unei aliaje într-o piesă utilizabilă depinde, de asemenea, de controlul procesului și de prelucrarea precisă prin mașini-unelte. De exemplu, Shaoyi Metal Technology aplică prelucrarea prin mașini-unelte certificată conform IATF 16949 și controlul calității bazat pe SPC pentru a sprijini trecerea pieselor metalice din faza de prototipuri la utilizarea în producție.
 Serii mici, standarde ridicate. Serviciul nostru de prototipare rapidă face validarea mai rapidă și mai ușoară —
Serii mici, standarde ridicate. Serviciul nostru de prototipare rapidă face validarea mai rapidă și mai ușoară —
