Z akých kovov sa skladá oceľ? Dekódujte špecifikácie ocele pred kúpou

Z čoho sa oceľ vyrába?
Z čoho sa oceľ vyrába – prehľad
Oceľ je predovšetkým železo, obsahuje uhlík ako nevyhnutnú nekovovú zložku a v závislosti od triedy môže obsahovať aj ďalšie zličovacie kovy.
Ak hľadáte, z akých kovov sa oceľ skladá, začnite s východiskovým kovom: železom. To odpovedá na jednoduchú verziu otázky, z akého kovu sa oceľ skladá. Menej zrejmou zložkou je uhlík. Oceľ sa neskladá výlučne z kovov, pretože uhlík je nevyhnutný a uhlík je nekov. V bežnom jazyku: z čoho sa oceľ vyrába? Je to zliatina železa a uhlíka, niekedy s pridaním ďalších prvkov na dosiahnutie špecifických vlastností. Britannica popisuje oceľ ako zliatinu železa a uhlíka s obsahom uhlíka do 2 percent.
- Železo je hlavný kov v oceli.
- Uhlík je nevyhnutný, avšak nie je kov.
- Niektoré triedy obsahujú pridané prvky, ako napríklad mangán, chróm, nikl alebo molybdén.
- Nie všetky ocele obsahujú chróm alebo nikl.
Stručná odpoveď na otázku, z akých kovov sa oceľ skladá
Ak sa opýtate, z čoho sa oceľ vyrába alebo z čoho sa oceľ skladá, univerzálna odpoveď začína železom a uhlíkom. Ďalej sa zloženie líši podľa druhu ocele. Uhlíková oceľ môže pozostávať predovšetkým z železa a uhlíka, zatiaľ čo nehrdzavejúca oceľ je samostatnou skupinou, ktorá obsahuje aspoň 11 percent chrómu, ako uvádza Service Steel . Preto by ste nemali predpokladať, že každá trieda ocele obsahuje chróm alebo nikel.
Prečo je uhlík dôležitý, aj keď nie je kovom
Čisté železo je relatívne mäkké. Malé množstvá uhlíka ho posilnia a premenia na oveľa užitočnejší technický materiál, čo potvrdzuje prehľad ocele v encyklopédii Britannica. Takže je oceľ zliatina? Áno. Je oceľ kov? V bežnom používaní áno, no technicky ide o skupinu železo-založených zliatin. Ak sa stále pýtate z čoho sa oceľ skladá , stručná odpoveď znie: z železa, uhlíka a niekedy aj iných prvkov. Ktoré prvky sú vždy prítomné, bežné, voliteľné alebo len stopové, to je oblasť, kde sa chémia stáva oveľa praktickejšou.
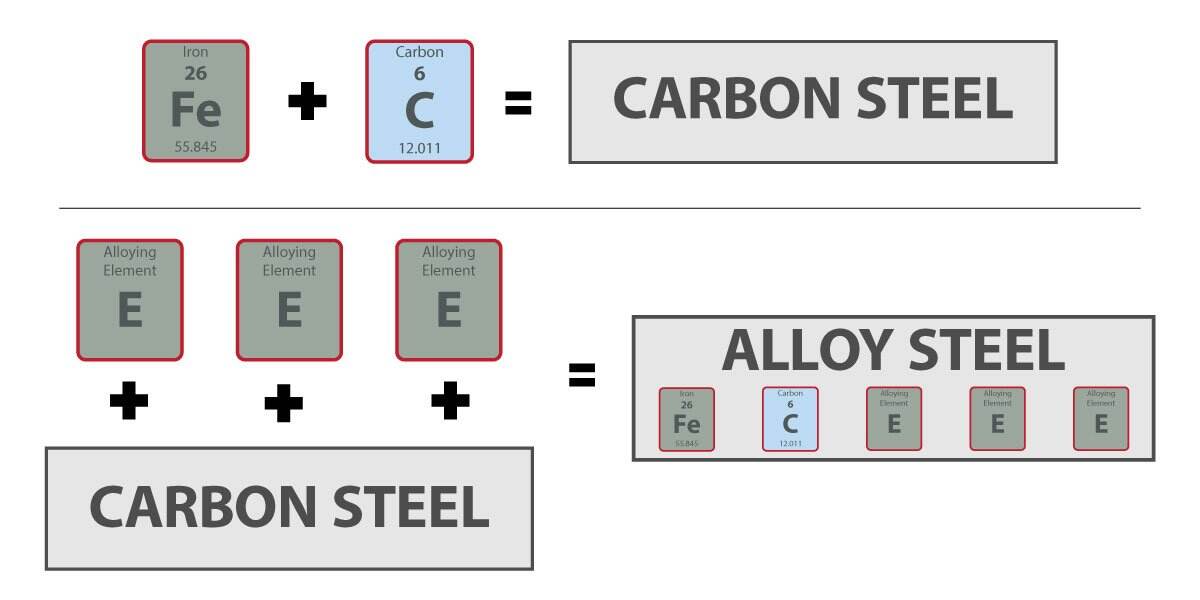
Ktoré prvky sa nachádzajú v oceli podľa kategórií
Chemická správa môže vyzerať preplnená, ale vzor je jednoduchší, než sa zdá. Zloženie ocele sa zvyčajne rozdeľuje do štyroch kategórií: vždy prítomné, bežné v mnohých triedach, niekedy pridané na konkrétny účel a stopové alebo zvyškové. Toto rozlíšenie je dôležité, pretože nie každý prvok uvedený v certifikáte ocele bol úmyselne pridaný a nie každý uvedený prvok ovplyvňuje výkon rovnakým spôsobom.
Základný kov a nevyhnutné zložky
Ak sa pýtate, či sa oceľ vyrába z železa, praktická odpoveď je áno, avšak nie iba z železa. Spoločnosť MISUMI popisuje oceľ ako zliatinu železa a uhlíka, pričom obsah uhlíka je zvyčajne nižší ako 2 percentá. Takže na najvyššej úrovni sa oceľ skladá z železnej základne plus uhlíka . Ak ste sa niekedy pýtali, ktorý iný prvok sa kombinuje s železom pri výrobe ocele, definujúcou odpoveďou je uhlík. Železo je základným kovom. Uhlík je nevyhnutný, avšak je to nemetal, preto úplný zoznam zložiek zahŕňa aj kovové, aj nemetalové prvky.
Bežné prísady na zliatiny a voliteľné kovy
Mnoho komerčných ocelí obsahuje tiež mangán a kremík. Bailey Metal Processing uvádza, že mangán je prítomný vo všetkých komerčných oceliach ako prísada, zvyčajne v rozmedzí približne 0,20 % až 2,00 %. Kremík môže byť buď úmyselnou prísadou, alebo zvyškovým prvkom, podľa triedy a výrobného procesu. Okrem toho sú voliteľné kovy, ako napríklad chróm, nikel, molybdén, vanád, niób a titán, špecifické pre konkrétne triedy. Tieto prvky sa pridávajú vtedy, keď oceľ vyžaduje cieľové vlastnosti, ako napríklad vyššiu pevnosť, lepšiu kaliteľnosť alebo zlepšenú odolnosť voči korózii. Inými slovami, oceľ sa skladá z základného zloženia plus prísad na optimalizáciu výkonu, ktoré sa líšia podľa rodiny ocelí.
| Kategória | Príkladové prvky | Prečo sa vyskytujú | Čo by čitateľ mal vyvodiť |
|---|---|---|---|
| Vždy prítomné | Železo, uhlík | Železo je základný kov. Uhlík definuje oceľ ako zliatinu železa s uhlíkom. | Toto je minimálna odpoveď na otázku, aké prvky obsahuje oceľ. |
| Bežné v mnohých komerčných oceliach | Mangán, kremík | Používa sa na bežnú kontrolu zloženia a úpravu vlastností v mnohých triedach ocele. | Oceľ pozostávajúca z železa, uhlíka, mangánu a kremíka ešte stále nie je automaticky nehrdzavejúca ani špeciálna oceľ. |
| Niekedy sa pridáva | Chróm, nikl, molybdén, vanád, niób, titán, bór, hliník, vápnik | Pridáva sa za účelom dosiahnutia špecifických výkonnostných cieľov, ako sú pevnosť, uzatvárateľnosť, kontrola veľkosti zrna, odkysličovanie alebo odolnosť voči korózii. | Presné zloženie závisí od triedy a predpokladaného použitia. |
| Stopové množstvá alebo zvyškové prvky | Fosfor, síra, meď, dusík, malé zvyškové množstvá niklu alebo chrómu | Vyskytujú sa náhodne z surovín alebo šrotu, alebo sa udržiavajú na kontrolovanej nízkej úrovni. | Uvedený prvok nie je vždy zámerným prísadovým prvkom zliatiny. |
Vysvetlenie zvyškových prvkov a nečistôt
Práve tu sa čitatelia často zamotajú. Bailey vysvetľuje, že niektoré prvky sa vyskytujú náhodne a nedajú sa ľahko odstrániť, preto sa považujú za stopové alebo zvyškové prvky. Fosfor je často zvyškový, síru sa zvyčajne zníži, pretože je všeobecne škodlivá, a zvyškové množstvá medi, niklu, chrómu a molibdénu sa kontrolujú prostredníctvom správy šrotu. Keď teda prečítate zloženie ocele, pamätajte si, že oceľ sa skladá z hlavnej štruktúry, bežných podporných prísad a pozadia chémie, ktoré môže, ale nemusí byť zámerné. Tým sa odpovedá na otázku týkajúcu sa kategórií. Výstižnejšou otázkou je však to, aký vplyv má každý z týchto prvkov v skutočnosti vo vnútri kovu.
Kovy v oceli a úloha jednotlivých prvkov
Ocelová trieda začne dávať viac zmyslu, keď prestanete čítať jej označenie ako náhodný zoznam symbolov a začnete ho čítať ako recept. Niektoré zložky ocele tvoria základnú štruktúru. Iné jemne upravujú správanie kovu v zváracom stredisku, strojníckom závode alebo korozívnom prevádzkovom prostredí. To je skutočná odpoveď na otázku zloženia ocelového kovu: každý prvok si zaslúži svoje miesto tým, že konkrétnym spôsobom mení výkon materiálu.
Železo a uhlík ako základ ocele
Železo je hlavným kovom v oceli. Jednoducho povedané, je to kostra, na ktorej sa všetko ostatné staví. Presnejšie povedané, oceľ je zliatina na báze železa a železo pôsobí ako matrica, ktorá udržiava uhlík a ďalšie legovacie prvky.
Uhlíka nie je kov, ale je najdôležitejším zliatinovým prvkom v ocele. V jazyku vhodnom pre začiatočníkov uhlík premieňa relatívne mäkké železo na oveľa pevnejší technický materiál. Z metalurgického hľadiska uhlík zvyšuje medzu pevnosti v ťahu, tvrdosť, odolnosť voči opotrebovaniu a schopnosť kalenia, zároveň však zníži tažnosť, húževnatosť, obrábateľnosť a zvárateľnosť. Pokyny od STI/SPFA uvádzajú, že obsah uhlíka v oceli môže dosiahnuť až 2 %, pričom väčšina zváraných ocelí obsahuje menej ako 0,5 % uhlíka.
Ak sa pýtate, z akých prvkov sa oceľ skladá, tieto dva sa uvádzajú vždy ako prvé: železo ako základný kov a uhlík ako nevyhnutný nemetal.
Zliatinové kovy, ktoré menia výkon
Mangan je bežný v mnohých triedach ocele. Jednoducho povedané, zvyšuje pevnosť ocele a zlepšuje jej spracovateľnosť počas výroby. Z technického hľadiska pôsobí ako odkysličovadlo, bráni tvorbe sírového železa a zvyšuje schopnosť kalenia a odolnosť voči opotrebovaniu. Podľa STI/SPFA ocele zvyčajne obsahujú aspoň 0,30 % mangánu, pričom niektoré uhlíkové ocele môžu obsahovať až 1,5 % mangánu.
Silikón sa často pridáva v malých množstvách na očistenie taveniny. Presnejšie povedané, ide o odkysličovadlo, ktoré môže zároveň zvýšiť pevnosť a tvrdosť. Kompenzáciou je, že vyššia výsledná pevnosť zváraného kovu môže byť spojená s nižšou tažnosťou a vyšším rizikom trhliny v niektorých prípadoch.
Chromu je jedným z najznámejších kovov v ocele, pretože zvyšuje odolnosť voči korózii, tvrdosť, schopnosť kalenia a odolnosť voči oxidácii pri vysokých teplotách. V nehrdzavejúcich zliatinách, ako uvádza STI/SPFA, obsah chrómu môže presiahnuť 12 %. Kompenzáciou je, že niektoré chrómové ocele sa okolo zvarov môžu stať tak tvrdými, že vznikajú trhliny.
Nikel pomáha oceli udržať si húževnatosť. Jednoduchým jazykom povedané, zvyšuje pevnosť bez toho, aby materiál stal príliš krehký. Technickejšie povedané, zlepšuje húževnatosť a tažnosť a je obzvlášť užitočná v prípadoch, keď je dôležitý výkon pri nízkych teplotách.
Molibdén pomáha oceli odolávať vysokým teplotám a zlepšuje jej schopnosť kalenia. Používa sa tiež na zvýšenie odolnosti voči puklinovému koroziu v niektorých nehrdzavejúcich oceliach. Rovnaké zdroje uvádzajú, že sa zvyčajne vyskytuje v zliatinových oceliach v množstve nižšom ako 1 %.
Vanádium používa sa v veľmi malých množstvách, avšak jej účinok je nepomerne veľký. Zvyšuje pevnosť, tvrdosť, odolnosť proti opotrebovaniu a odolnosť proti nárazu a pomáha kontrolovať rast zŕn. Kompenzáciou je to, že v vyšších koncentráciách môže prispieť k krehknutiu počas tepelnej úpravy.
Malé prísady s veľkými metalurgickými účinkami
Nie každý prvok uvedený v správe je tam preto, aby ocel zlepšil vo všetkých ohľadoch. Niektoré prvky sa kontrolujú, pretože ich účinok je prospešný len v obmedzených prípadoch. Sír má za úlohu zlepšiť obrábateľnosť v oceliach určených na ľahké obrábanie, avšak znižuje ich zvárateľnosť, tažnosť a nárazovú húževnatosť. Fosfor môže zvýšiť pevnosť a obrábateľnosť a zároveň zvyšuje krehkosť. Hliník sa často pridáva v veľmi malých množstvách ako odkysličovadlo a jemnozrnný prísadový prvok na zlepšenie húževnatosti. Preto sa kovové prísady v ocele najlepšie chápu ako súbor kompromisov, nie ako zoznam automatických vylepšení.
| Prvkový | Kov alebo nekov | Hlavný účinok v oceli | Bežné rodiny ocelí | Kľúčový kompromis |
|---|---|---|---|---|
| Železo | Kov | Základná matrica zliatiny | Všetky ocele | Čisté železo je samotné relatívne mäkké |
| Uhlíka | Nekrystalické materiály | Zvyšuje tvrdosť, pevnosť, odolnosť voči opotrebovaniu a schopnosť kalenia | Všetky ocele, najmä uhlíkové a nástrojové ocele | Nižšia zvárateľnosť, ťahová pevnosť, húževnatosť, obrábateľnosť |
| Mangan | Kov | Odstraňuje kyslík, zvyšuje pevnosť a schopnosť kalenia | Mnoho uhlíkových a zliatinových ocelí | Vyššia tvrdosť môže komplikovať tvárnenie alebo zváranie |
| Silikón | Nekrystalické materiály | Odstraňuje kyslík a zvyšuje pevnosť | Mnoho komerčných ocelí, zváracích materiálov, liatych ocelí | Príliš veľké množstvo môže znížiť ťahovú pevnosť |
| Chromu | Kov | Zlepšuje odolnosť voči korózii, tvrdosť a schopnosť kalenia | Nerezové, zliatinové a nástrojové ocele | Môže zvýšiť tvrdosť zváracích zón a riziko vzniku trhlin |
| Nikel | Kov | Zvyšuje húževnatosť a pevnosť | Zliatiny ocele, niektoré nehrdzavejúce ocele | Nie je prítomný vo všetkých triedach nehrdzavejúcich ocelí |
| Molibdén | Kov | Zvyšuje schopnosť kalenia a pevnosť pri vyšších teplotách | Zliatiny ocele, niektoré nehrdzavejúce ocele | Zvyšuje náklady a môže komplikovať výber spracovateľských metód |
| Vanádium | Kov | Zvyšuje pevnosť, odolnosť proti opotrebovaniu a kontrolu zrna | Vysokopevnostné nízkolegované ocele (HSLA), nástrojové ocele, zliatiny ocele | Vyššie množstvá môžu prispieť k krehknutiu |
| Sír | Nekrystalické materiály | Zlepšuje obrábateľnosť v triedach ocelí s vylepšenou obrábateľnosťou | Ocele s pridaným sírou | Znižuje zvárateľnosť a húževnatosť |
| Fosfor | Nekrystalické materiály | Môže zvyšovať pevnosť a obrábateľnosť | Zvyčajne sa v uhlíkových oceliach udržiava na nízkej úrovni | Zvyšuje krehkosť |
| Hliník | Kov | Odstraňovač kyslíka a jemnozrnný prísadový prvok | Jemnozrnné ocele | Zvyčajne je užitočný len v veľmi malých množstvách |
Ak sa na to pozrieme takto, otázka, aké prvky tvoria oceľ, je len polovicou celého problému. Druhou polovicou je otázka, či je oceľ jednou látkou, prvkom alebo niečím zložitejším, než naznačuje prvý zoznam zložiek.
Je oceľ prvkom, zlúčeninou alebo zmesou?
Zoznam zložiek vám hovorí, čo do ocele vstupuje. Chémia však kladie inú otázku: aký druh látky to je? Oceľ nie je prvok, preto sa na periodickej tabuľke neobjavuje ako samostatný prvok. Nemá ani žiadny jednotný chemický symbol pre oceľ a ani žiadny jednotný chemický vzorec pre oceľ. Sciencing upozorňuje, že chemický vzorec ocele nie je pevne stanovený, pretože oceľ je zmesou, presnejšie zliatinou železa a uhlíka, ktorá môže obsahovať aj ďalšie prvky v závislosti od triedy.
Prečo oceľ nemá chemickú značku
Oceľ je zliatina, nie chemický prvok, a preto nemá jedinečnú značku ani pevný molekulárny vzorec.
- Mýtus: Oceľ má značku ako Fe. Fakt: Fe je značka pre železo, nie pre oceľ.
- Mýtus: Oceľ by mala mať jeden vzorec. Fakt: Rôzne triedy používajú rôzne zloženia, preto sa žiadny jediný vzorec na všetky nezhoduje.
- Mýtus: Oceľ je zlúčenina ocele. Fakt: V metalurgii sa klasifikuje ako zliatina, nie ako jedna pevne daná zlúčenina.
Oceľ vs železo v periodickej tabuľke
Ak ste sa niekedy pýtali, či je oceľ prvkom alebo či sa oceľ nachádza v periodickej tabuľke, odpoveď na obe otázky je nie. Periodická tabuľka obsahuje len čisté prvky, ako napríklad železo, chróm a nikl. Oceľ sa vyrába z prvkov, avšak sama o sebe nie je chemickým prvkom. Wikipedia popisuje oceľ ako zliatinu železa a uhlíka, pričom do mnohých tried sa pridávajú aj ďalšie prvky.
Zliatina, zmes alebo zlúčenina?
Ak sa pýtate, či je oceľ zlúčenina alebo zmes, krátka odpoveď je: v bežnom jazyku zmes a v technickom jazyku zliatina. Zlúčenina má pevné chemické zloženie, napríklad voda. Oceľ nie. Jej chemické zloženie sa mení od triedy k triede, preto hľadanie chemického vzorca pre oceľ nevedie nikam užitočného. Zvonka môže vyzerať jednotne, avšak jej vnútorná mikroštruktúra môže byť zložitejšia, pričom rôzne fázy vznikajú v závislosti od zloženia a tepelnej úpravy. Preto sa uhlíková oceľ, nehrdzavejúca oceľ, legovaná oceľ a nástrojová oceľ všetky môžu nazývať oceľou, hoci sa v praxi správajú veľmi odlišne.

Zloženie rodiny ocelí
Tieto rodinné názvy sú viac než len zjednodušené označenia používané na výrobnej ploche. Oznamujú vám, ktoré zložky v recepte prevládajú. Keď kupujúci položia otázku, z akých kovov sa vyrába oceľ, odpoveď závisí od toho, ktorú rodinu ocele majú na mysli. Medzi hlavnými typmi ocele uhlíková oceľ je najbližšie železu s pridaným uhlíkom, nehrdzavejúca oceľ je definovaná prítomnosťou chrómu, zliatiny ocele obsahujú pridané prvky na prispôsobenie výkonu a nástrojová oceľ dosahuje vyššiu tvrdosť a odolnosť proti opotrebovaniu prostredníctvom vyššieho obsahu uhlíka a špeciálnych zliatinových prísad.
Zloženie uhlíkovej ocele a vysokouhlíkovej ocele
Medzi rôznymi typmi ocele je uhlíková oceľ najjednoduchšie pochopiteľná z hľadiska chémie. Uhlík v uhlíkovej oceli je hlavným kritériom triedenia, nie chróm ani nikl. Bežné klasifikácie zhrnul TWI a BigRentz umiestnite nízkouhlíkovú oceľ s obsahom uhlíka približne 0,25 až 0,30 %, stredne uhlíkovú oceľ s obsahom uhlíka približne 0,25 až 0,60 % a vysokouhlíkovú oceľ s obsahom uhlíka približne 0,60 až 1,25 %; presné hranice sa však líšia podľa zdroja a normy. So zvyšujúcim sa obsahom uhlíka sa zvyšuje aj tvrdosť a odolnosť voči opotrebovaniu. Škvrnitosť, tvárnosť a zvárateľnosť sa zvyčajne menia opačným smerom. Preto sa nízkouhlíkové triedy často používajú pri tvarovaných a zváraných súčiastkach, zatiaľ čo vyššie uhlíkové triedy sa používajú tam, kde je dôležitejšia tuhosť, udržanie ostrosti rezného okraja alebo odolnosť voči opotrebovaniu.
Prečo nehrdzavejúca oceľ obsahuje rôzne legovacie kovy
Rozdiel medzi uhlíkovou a nehrdzavejúcou oceľou je v skutočnosti rozdiel v chémii. Nehrzdávajúca oceľ musí obsahovať aspoň 10,5 % chrómu, ako uvádza TWI, a práve tento chróm poskytuje tejto skupine jej koróznu odolnosť. Nikel je bežný v mnohých triedach nehrdzavejúcej ocele, najmä v austenitickej nehrdzavejúcej oceli, avšak nie je všadeprítomný. Ferritické nehrdzavejúce ocele často obsahujú len veľmi malé množstvo niklu alebo vôbec žiadny nikl. Nikel Institute vysvetľuje, že nikel zlepšuje tvarovateľnosť, zvárateľnosť, kujnosť a odolnosť voči korózii v mnohých triedach nehrdzavejúcej ocele, čo je dôvod, prečo sa nehrdzavejúca oceľ obsahujúca nikel tak široko používa. Napriek tomu je chróm tým prvkom, ktorý definuje nehrdzavejúcu oceľ. Nikel zlepšuje výkon niektorých druhov nehrdzavejúcej ocele.
Ako sa zapájajú legované ocele a nástrojové ocele
Legovaná oceľ predstavuje širokú strednú kategóriu. Stále ide o železo-uhlíkovú oceľovú zliatinu, avšak s úmyselnými prísadami, ako sú napríklad mangán, molybdén, chróm, nikel, kremík alebo vanád, ktoré zameriavajú vlastnosti na zlepšenie schopnosti kalenia, pevnosti, húževnatosti alebo odolnosti voči teplu. Nástrojová oceľ ide ešte ďalej. Spoločnosť BigRentz popisuje nástrojovú oceľ ako rodinu vysokouhlíkových ocelí určených na výrobu nástrojov, ktoré sa často zvyšujú pevnosťou pomocou prvkov, ako sú chróm, tungsten, vanád a molybdén. Hoci sú všetky ocele technicky zliatinami, pojem „legovaná oceľ“ ako samostatná kategória zvyčajne označuje materiál, ktorý je viac technicky navrhnutý v porovnaní s bežnou uhlíkovou oceľou, pričom nástrojová oceľ predstavuje špeciálny koniec tohto spektra.
| Rodina ocelí | Základné prvky | Definujúca chemická vlastnosť | Typické výhody | Bežné kompromisy |
|---|---|---|---|---|
| Uhlíková oceľ | Železo + uhlík, zvyčajne s obmedzenými pridanými zliatinovými prvkami | Klasifikované predovšetkým podľa obsahu uhlíka | Široko dostupné, cenovo výhodné, nízkouhlíkové triedy sa dobre tvária a zvárajú, vyššie uhlíkové triedy získavajú tvrdosť | Nižšia odolnosť voči korózii v porovnaní s nehrdzavejúcimi ocelami a vyšší obsah uhlíka komplikuje spracovanie |
| Zliatinová oceľ | Železo + uhlík + pridané prvky, ako napríklad mangán, chróm, nikl, molybdén, kremík alebo vanád | Zloženie je upravené tak, aby sa dosiahli požadované mechanické alebo tepelné vlastnosti | Možno prispôsobiť pevnosť, schopnosť kalenia, húževnatosť a výkon pri vysokých teplotách | Špecifikácie sa stávajú zložitejšími a náklady aj nároky na spracovanie často stúpajú |
| Nehrdzavejúca oceľ | Železo + uhlík + najmenej 10,5 % chrómu, pričom mnoho tried obsahuje aj nikl | Chróm definuje túto skupinu a zabezpečuje odolnosť voči korózii | Lepšia odolnosť voči korózii, trvanlivosť a v niektorých triedach aj vysoká tvárnosť a čistota | Zvyčajne vyššia cena a odolnosť voči korózii a magnetizmus sa líšia podľa podtypu |
| Nástrojová ocele | Železná oceľ s vyšším obsahom uhlíka s legovacími prvkami, ako sú chróm, wolfrám, vanád alebo molybdén | Navrhnutá pre extrémnu tvrdosť, odolnosť proti opotrebovaniu a udržanie ostrosti hrany | Výborná pre diely, rezače, vrtáky a iné náročné nástroje | Nižšia tažnosť, ťažšie obrábanie a náročnejší výber tepelného spracovania |
Ak sa rôzne typy ocele porovnajú vedľa seba, prestanú pripomínať váhavo definované kategórie a začnú pôsobiť ako rozhodnutia z oblasti chémie. Malá zmena obsahu uhlíka, chrómu alebo niklu môže rozhodnúť, či sa daná trieda ľahko zvára, odoláva hrdze, čisto obrába alebo vydrží opakované opotrebovanie.
Ako sa zloženie ocele ovplyvňuje jej výkon
Tieto voľby zloženia sa rýchlo prejavia v reálnom použití. Malá zmena obsahu uhlíka, chrómu, niklu, molybdénu alebo síry môže ovplyvniť, či sa oceľ dobre opotrebuje, odoláva korózii, spracováva čistou rezaním alebo spôsobuje problémy počas výroby.
Ako prvky ovplyvňujú pevnosť a tvrdosť
Diehl Steel popisuje uhlík ako najdôležitejšiu zložku ocele. V praxi väčší obsah uhlíka zvyčajne znamená vyššiu medzu pevnosti v ťahu, tvrdosť a odolnosť proti opotrebovaniu a abrázii. Za túto výhodu sa platí nižšou tažnosťou, húževnatosťou a obrábateľnosťou. Chróm tiež zvyšuje pevnosť, tvrdosť, schopnosť kalenia a odolnosť proti opotrebovaniu. Molybdén zvyšuje pevnosť a schopnosť kalenia a pomáha oceli udržať jej vlastnosti pri vyšších teplotách. Nikel je obzvlášť užitočný, pretože zvyšuje pevnosť a tvrdosť bez toho, aby tak výrazne znížil tažnosť a húževnatosť.
- Uhlík: lepšia tvrdosť a odolnosť proti opotrebovaniu, ale nižšia schopnosť ohybu a natiahnutia.
- Chróm a molybdén: silnejšia odpoveď na kalenie a náročné prevádzkové podmienky.
- Nikél: extra sila s užitočnou odolnosťou.
Prečo niektoré ocele odolávajú korózii lepšie ako iné
Ak sa pýtate, či oceľ hrdzavie, mnohé ocele áno. Skutočnou otázkou je, či odolnosť voči korózii vyplýva z zloženia zliatiny samotnej alebo z ochranného povrchového vrstvy. Diehl uvádza, že chróm zvyšuje odolnosť voči korózii, a preto sa nehrdzavejúce ocele správajú inak ako bežné uhlíkové ocele. V galvanizovaná vs Nezardobná oceľ porovnaní Tuhé životné linky vysvetľuje, že pozinkovaná oceľ je uhlíková oceľ chránená zinkovým povlakom, zatiaľ čo nehrdzavejúca oceľ je zliatina železa, chrómu a ďalších prvkov odolných voči korózii. Inými slovami, ochrana pozinkovania sa nachádza na vonkajšej strane, zatiaľ čo vlastnosti nehrdzavejúcej ocele sú do materiálu zabudované.
- Nerezová ocel: odolnosť voči korózii vyplýva zo zloženia.
- Galvanizovaná oceľ: ochrana pred koróziou vyplýva zo zinkového povlaku.
- Oceľ vs. železo: oceľ vychádza z železa, avšak pridané prvky menia jej výkon v prevádzke.
Kompromisy medzi zvárateľnosťou, obrábateľnosťou a odolnosťou
Niektoré prísady pomáhajú jednému výrobnému kroku, ale zároveň škodia inému. Sír je najjasnejším príkladom. Diehl uvádza, že sír zlepšuje obrábateľnosť vo voľne obrábaných oceliach, avšak znižuje zvárateľnosť, nárazovú húževnatosť a tažnosť. Priemyselní metalurgovia uvádzajú, že sír sa spája s mangánom za vzniku inklúzií sulfidu mangánového, ktoré pomáhajú pri obrábaní lomiť triesky. Práve tieto inklúzie sú dôvodom, prečo sa voľne obrábané ocele môžu ťažko zvárať, najmä keď je obsah síra a fosforu zvýšený.
- Pre obrábanie: sír môže zlepšiť kontrolu triesok.
- Pre zváranie: vyšší obsah síra negatívne ovplyvňuje kvalitu zvarov.
- Pre húževnatosť: nikel podporuje húževnatosť, kým sír a fosfor posúvajú oceľ smerom k krehkosti.
Preto údaj o zložení v certifikáte materiálu nie je len laboratórnym údajom. Je to predbežná informácia o správaní materiálu v dielni a o výkone výrobku, čo sa stáva výrazne jasnejšie, ak viete, ako samotnú technickú špecifikáciu čítať.

Ako čítať správy o zložení ocele
Certifikát výrobcu môže pôsobiť ako stena skratiek. Ak ho čítate vrstvami, stane sa oveľa jednoduchším. Pre kupujúcich, študentov a výrobcov nie je cieľom naučiť sa každý kód nazubom, ale overiť zloženie ocele, ktorú ste objednali. Typický protokol o skúške výrobcu (MTR) spája materiál s číslom tavby a uvádza chemické zloženie, mechanické vlastnosti, splnené normy, rozmery, povrchovú úpravu a overovací podpis.
Ako prečítať protokol o chemickom zložení
- Najskôr porovnajte číslo tavby. Toto prepojí protokol s konkrétnou dávkou kovu a poskytne vám možnosť sledovania.
- Nájdite časť týkajúcu sa chemického zloženia ocele. Hľadajte symboly prvkov, napríklad C, Mn, Cr a Ni, spolu s ich percentuálnymi hodnotami.
- Skontrolujte povolené rozsahy. Niektoré tabuľky uvádzajú minimálne a maximálne limity. MD Metals poznámka: Tieto rozsahy definujú prijateľné okno chemického zloženia pre danú triedu.
- Oddeľte chemické zloženie od výsledkov skúšok. Pevnosť v ťahu, mezná pevnosť a predĺženie, ako aj tvrdosť opisujú výkon pri skúškach, nie samotné zložky.
- Všimnite si znaky výroby. Ak sa objaví uhlíková ekvivalencia, považujte ju za signál zvárateľnosti. Vyššia hodnota CE môže znamenať náročnejšie podmienky zvárania.
Na čo si treba dávať pozor v popisoch tried
Riadok s triedou vám hovorí, podľa ktorej pravidelnej knihy sa riadi. Protokol o materiálových vlastnostiach (MTR) môže odkazovať na požiadavky ASTM, ASME alebo SAE, zatiaľ čo tabuľka zloženia ukazuje skutočné chemické zloženie ocele v danom konkrétnom tave. Toto rozlíšenie je dôležité. Názov triedy vám hovorí, čomu musí oceľ vyhovovať. Tabuľka prvkov ukazuje, kde sa dodaná dávka nachádza v rámci týchto limít. Ak je uvedené Fe, MD Metals upozorňuje, že sa môže objaviť ako minimálna hodnota, zatiaľ čo uhlík a zliatinové prísady sa zvyčajne uvádzajú v percentách.
Ako rozlíšiť základnú chemickú zložku od povrchových povlakov
Zloženie ocele patrí do tabuľky chémie. Veľkosť výrobku, hrúbka a povrchová úprava patria na iné miesto. Mill Steel oddeľuje chemické zloženie od rozmerov a popisu výrobku, čo je užitočný zvyk pri čítaní akéhokoľvek certifikátu. Ak dokument spomína povrchovú úpravu alebo popis pozinkovaného výrobku, nezamieňajte túto poznámku s chemickým zložením základnej zliatiny.
| Políčko správy | Čo to znamená | Prečo je to dôležité |
|---|---|---|
| Počet tepla | Jedinečný identifikátor šarže | Potvrdzuje sledovateľnosť |
| Chemické zloženie | Symboly prvkov a ich percentuálne podiely | Ukazuje zloženie samotnej ocele |
| Mechanické vlastnosti | Údaje o pevnosti, tvrdosti a predĺžení | Ukazuje výsledky skúšok, nie chemické zloženie |
| Dodržané špecifikácie | Odkazované normy alebo trieda | Uvádza, ktoré požiadavky sa uplatňujú |
| Rozmery a povrchová úprava | Veľkosť, hrúbka, popis výrobku | Udržiava podrobnosti povrchu oddelené od základného zloženia materiálu |
| Podpis osoby oprávnenej na certifikáciu | Autorizácia výrobného závodu | Potvrdzuje, že správa je certifikovaná |
Ak takto prečítate certifikát, začnú sa dokumenty skutočne používať. Stávajú sa praktickým nástrojom na posúdenie toho, či daná oceľ vyhovuje danej aplikácii, technologickému procesu a otázkam, ktoré by ste mali položiť ešte pred výrobou súčiastok.
Vyberte správny druh ocele pre tažené súčiastky
Zloženie ocele je najdôležitejšie vtedy, keď ovplyvní konkrétne rozhodnutie. Ak viete, z akej ocele sú vyrobené súčiastky vo vašej zostave, môžete si položiť presnejšie otázky týkajúce sa tvárnosti, pevnosti, ochrany proti korózii a nákladov už pred zahájením výroby nástrojov. Mill Steel jasne zdôrazňuje základné priority pri tažení: tvárnosť, povrchovú úpravu, úzke tolerancie hrúbky, predvídateľné mechanické vlastnosti a – ak je to potrebné – povrchové povlaky na ochranu proti korózii. QST pridáva praktické filtre, s ktorými sa zvyčajne stretávajú nakupujúci, vrátane trvanlivosti, hrúbky, tvrdosti, odolnosti voči korózii a konzistentnosti dodávateľa.
Prispôsobiť chemické zloženie ocele funkcií súčiastky
Ľudia sa často pýtajú, na čo sa oceľ používa, alebo dokonca zadajú do vyhľadávacieho poľa výraz „na čo sa oceľ používa“, akoby existovala jediná odpoveď. Pri tvárnení sa z ocele vyrábajú súčiastky od jednoduchých upevňovacích prvkov a krytov až po karosérie automobilov, posilovacie prvky a podvozkové súčiastky. Nízkouhlíkové a tažné triedy ocele sa zvyčajne vyberajú v prípadoch, keď je potrebné jednoduchšie tvárnenie súčiastky. Vysokopevnostné nízkolegované (HSLA) ocele sú vhodné, keď tenšia plechová polotovar musí stále prenášať väčšie zaťaženie. Galvanicky pozinkovaný plech je užitočný v prípadoch, keď ochrana proti korózii pochádza z cinkového povlaku, nie z chemického zloženia základnej zliatiny.
Otázky, ktoré sa máte na výrobcu opýtať týkajúce sa výberu ocele
- Ktorá oceľová trieda najlepšie zodpovedá tvaru, zaťaženiu a prevádzkovému prostrediu súčiastky?
- Potrebujeme jednoduchšie tvárnenie, vyššiu pevnosť alebo lepšiu odolnosť voči korózii?
- Bude lepším riešením nízkouhlíková oceľ, tažná oceľ, HSLA oceľ, nehrdzavejúca oceľ alebo pozinkovaný plech?
- Pochádza ochrana proti korózii z chemického zloženia ocele alebo z povrchovej vrstvy?
- Spôsobí hrúbka, tvrdosť alebo zvárateľnosť problémy s nástrojmi alebo montážou?
- Môže dodávateľ zabezpečiť opakovateľnú kvalitu, sledovateľnosť a certifikáciu v rámci všetkých výrobných sérií?
Praktický zdroj informácií pre projekty automobilového tvárnenia
Tieto otázky nadobúdajú ešte väčší význam v automobilovom priemysle, kde rôzne druhy ocele môžu ovplyvniť hmotnosť, tuhosť, správanie sa pri zváraní a trvanlivosť. Ak potrebujete okrem diskusie o materiáloch aj podporu pri výrobe, Shaoyi je jedným z praktických zdrojov, ktoré stojí za zváženie. Shaoyi je dôverovaný viac ako 30 automobilovým značkám po celom svete a vyrába presne technicky navrhnuté diely pre automobilové tvárnenie v akomkoľvek výrobnom rozsahu. Jeho proces certifikovaný podľa štandardu IATF 16949 pokrýva všetko – od rýchleho prototypovania po automatizovanú sériovú výrobu dielov, ako sú riadiace ramená a podvozkové rámy. Pre nakupujúcich, ktorí sa rozhodujú, aký druh ocele určiť, takáto výrobná diskusia pomáha prepojiť zloženie zliatiny s dielom, ktoré sa skutočne dá vyrobiť, skontrolovať a dodávať s dôverou.
Často kladené otázky o zložení ocele
1. Aké kovy obsahuje oceľ?
Železo je hlavným kovom v oceli. Mnoho tried tiež obsahuje kovy, ako sú mangán, chróm, nikl, molybdén alebo vanád, avšak tieto prísady závisia od rodiny ocele a jej predpokladaného použitia. Úplná odpoveď zahŕňa aj uhlík, ktorý je pre oceľ nevyhnutný, hoci nie je kovom.
2. Je uhlík kovom v oceli?
Nie. Uhlík je nemetal, avšak je to práve tá zložka, ktorá mení železo na oceľ namiesto bežného železa. Aj mierny rozdiel v obsahu uhlíka môže ovplyvniť tvrdosť, odolnosť voči opotrebovaniu, tvárnosť, zvárateľnosť a húževnatosť, a preto má rovnako veľký význam ako kovové zliatiny.
3. Obsahujú všetky ocele chróm alebo nikl?
Nie. Mnoho bežných uhlíkových ocelí nepoužíva chróm ani nikl ako úmyselné zliatinové prísady. Nerezové ocele sú definované prítomnosťou chrómu, pričom nikl sa vyskytuje v mnohých nerezových triedach, avšak nie je všadeprítomný; preto by ste nemali predpokladať, že každá oceľ obsahuje oba tieto prvky.
4. Je oceľ prvok, zlúčenina alebo zmes?
Oceľ je najlepšie popísať ako zliatinu, čo je typ zmesi pozostávajúcej z železa, uhlíka a niekedy aj iných prvkov. Nie je to čistý prvok, nevyskytuje sa v periodickej tabuľke ako samostatný záznam a nemá ani jediný chemický symbol ani pevný chemický vzorec, pretože rôzne triedy ocele majú odlišné chemické zloženia.
5. Ako môžem zistiť, aké zloženie má konkrétna trieda ocele pred zakúpením súčiastok?
Začnite s certifikátom materiálu alebo skúšobným protokolom výrobcu. Skontrolujte číslo tavby, prečítajte si časť týkajúcu sa chémie – symboly prvkov a ich percentuálne zastúpenie – a oddeľte základnú zložku zliatiny od povlakov alebo dokončovacích úprav. Pri vyrazených automobilových súčiastkach je to obzvlášť užitočné, pretože dodávatelia, ako napríklad Shaoyi, dokážu pri výbere materiálu prepojiť prototypovanie, výrobné množstvá a požiadavky na kvalitu, keď voľba ocele ovplyvňuje tvárnosť, pevnosť alebo odolnosť voči korózii.
 Malé dávky, vysoké štandardy. Naša služba rýchlejho prototypovania urobí overenie rýchlejšie a jednoduchšie —
Malé dávky, vysoké štandardy. Naša služba rýchlejho prototypovania urobí overenie rýchlejšie a jednoduchšie —
