Kim Loại Được Cấu Tạo Từ Gì? Câu Trả Lời Đơn Giản Cùng Với Khoa Học Thực Tế
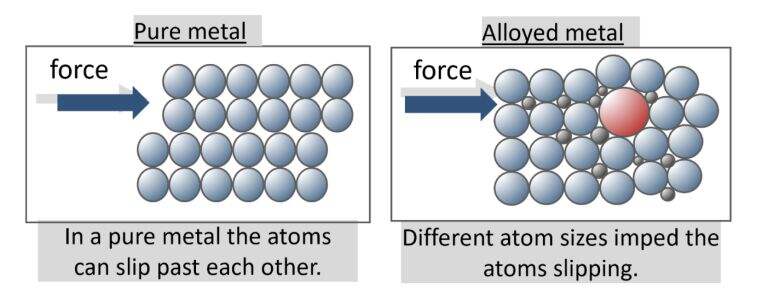
Câu trả lời trực tiếp cho câu hỏi kim loại được cấu tạo từ gì
Nếu bạn từng thắc mắc kim loại được cấu tạo từ gì, thì câu trả lời ngắn gọn phụ thuộc vào việc bạn hiểu 'kim loại' theo nghĩa nào: một nguyên tố, một nguồn tự nhiên hay một vật liệu có thể sử dụng được.
Kim loại có thể mang ba ý nghĩa liên quan đến nhau: một chất được cấu tạo từ các nguyên tử kim loại, một vật liệu được chiết xuất từ quặng trong lòng đất, hoặc một vật liệu thành phẩm có thể là kim loại nguyên chất hoặc hợp kim.
Kim loại được cấu tạo từ gì — giải thích đơn giản
Một cách diễn đạt đơn giản, kim loại được cấu tạo từ các nguyên tử của các nguyên tố kim loại như sắt, đồng hoặc nhôm. Trong tự nhiên, những nguyên tố này thường không tồn tại dưới dạng thanh hoặc tấm kim loại sạch sẵn sàng để sử dụng; thay vào đó, chúng thường bị 'khóa' bên trong quặng và khoáng vật, và phải được khai thác, tinh luyện. Trong đời sống hằng ngày, kim loại mà bạn chạm vào thường là một vật liệu đã qua chế biến, chứ không chỉ đơn thuần là một nguyên tố nguyên chất.
Đó là lý do vì sao những câu hỏi như kim loại được cấu tạo từ gì , kim loại được làm từ gì, hay thậm chí kim loại cấu tạo từ cái gì nghe có vẻ đơn giản nhưng lại có thể dẫn đến những câu trả lời khác nhau.
Ba cách trả lời đúng cho câu hỏi kim loại được cấu tạo từ gì
Có ba cách trả lời đúng cho câu hỏi này.
- Trong hóa học, kim loại được cấu tạo từ các nguyên tử kim loại sắp xếp trong một cấu trúc rắn.
- Trong tự nhiên, kim loại có thể sử dụng thường xuất phát từ quặng — loại vật liệu chứa các hợp chất mang kim loại.
- Trong sản xuất, một vật thể kim loại có thể được chế tạo từ kim loại nguyên chất hoặc từ hợp kim — một hỗn hợp được thiết kế nhằm cải thiện hiệu năng.
Britannica lưu ý rằng phần lớn kim loại được tìm thấy trong quặng, trong khi một số ít, như vàng hoặc đồng, có thể tồn tại ở dạng tự do.
Nguyên tử kim loại so với sản phẩm kim loại
Đây là sự phân biệt then chốt mà người mới thường bỏ qua. Một nguyên tử kim loại là một phần của một nguyên tố hóa học. Còn một sản phẩm kim loại — ví dụ như bu-lông thép hay chảo nhôm — là một vật phẩm đã qua chế tạo, được làm từ vật liệu kim loại. Vì vậy, khi ai đó hỏi 'kim loại được cấu tạo từ gì?', họ có thể đang đề cập đến nguyên tử, khai thác quặng hoặc các sản phẩm hoàn chỉnh.
Khoảng cách nhỏ về cách diễn đạt này chính là nơi khoa học thực sự bắt đầu, bởi vì câu trả lời sẽ thay đổi khi bạn chuyển từ cấp độ nguyên tử sang cấu trúc và cuối cùng là các vật liệu mà con người thực tế sử dụng.
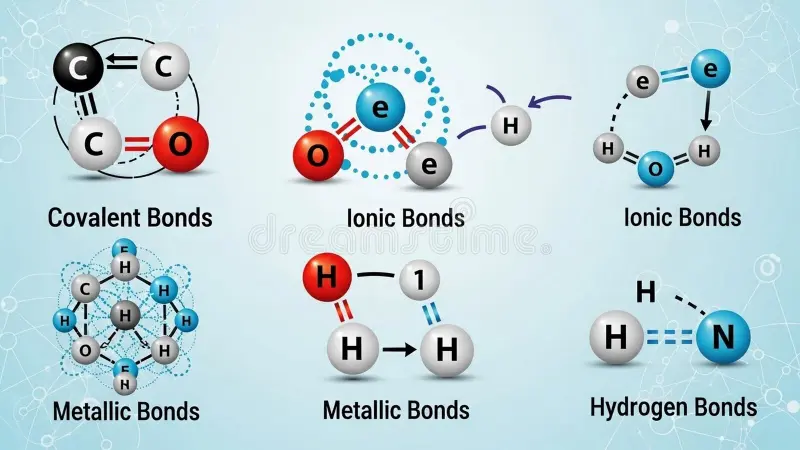
Liên kết kim loại hình thành nên các tính chất của kim loại
Câu trả lời bằng ngôn ngữ đơn giản là hữu ích, nhưng kim loại sẽ dễ hiểu hơn nhiều khi bạn phóng to đến cấp độ nguyên tử. Một thanh đồng, một tấm nhôm hoặc một mảnh sắt không thể hiện tính chất như vậy một cách ngẫu nhiên. Cấu trúc của chúng mang lại những đặc tính kim loại quen thuộc đó.
Điều gì làm cho một chất trở thành kim loại
Trong hóa học, một kim loại tinh khiết là một chất rắn tinh thể. Điều đó có nghĩa là các nguyên tử của nó được sắp xếp theo một mẫu đều đặn, lặp lại, chứ không tồn tại dưới dạng những phân tử nhỏ riêng biệt. LibreTexts giải thích rằng mỗi điểm trong mạng tinh thể này đều được chiếm bởi một nguyên tử giống hệt nhau, trong khi BBC Bitesize mô tả cấu trúc này là các ion kim loại được xếp chặt khít nhau theo những lớp đều đặn.
Sự sắp xếp đó là một phần lớn trong câu trả lời cho câu hỏi: kim loại có những tính chất gì? Kim loại không chỉ đơn thuần là những nguyên tử đứng yên. Chúng tạo thành một cấu trúc khổng lồ, trong đó các electron ở lớp ngoài cùng không bị gắn chặt vào một nguyên tử duy nhất như thường thấy ở nhiều chất khác.
Liên kết kim loại và hành vi của electron
Đây là cốt lõi của khái niệm kim loại trong hóa học. Trong một kim loại, các nguyên tử có thể được xem như những ion kim loại mang điện tích dương, bao quanh bởi các electron hóa trị linh động. Những electron linh động này được gọi là các electron phi cục bộ vì chúng có thể di chuyển xuyên suốt cấu trúc thay vì chỉ thuộc về một nguyên tử duy nhất. Liên kết kim loại là lực hút giữa các ion dương và đám mây electron chung đó.
Hãy hình dung nó như một khung sườn được xếp chặt, gắn kết với nhau nhờ các electron có khả năng di chuyển xuyên suốt vật liệu. Đó chính là lý do vì sao tính chất của kim loại cảm nhận được khác biệt so với muối, gốm sứ hay các chất phân tử.
Tại sao cấu trúc kim loại tạo ra những tính chất quen thuộc
Cách tốt nhất để hiểu các tính chất của kim loại là liên hệ từng tính chất ấy trở lại với cấu trúc của chúng.
- Điện dẫn và Nhiệt dẫn :các electron linh động có thể di chuyển qua kim loại và dẫn truyền điện tích cũng như năng lượng.
- Tính dẻo và tính kéo dài: các lớp trong mạng tinh thể có thể trượt lên nhau trong khi đám mây electron vẫn giữ cho cấu trúc nguyên vẹn.
- Bóng: ánh sáng tương tác với các electron trên bề mặt, giúp kim loại phản xạ và phát lại ánh sáng theo cách bóng loáng.
LibreTexts sử dụng một phép so sánh hữu ích: một tấm đồng có thể được định hình và rèn dập, nhưng đồng(I) clorua, dù cũng chứa đồng, lại bị vỡ thành dạng bột nếu xử lý theo cách tương tự. Vì vậy, khi người ta hỏi điều gì làm nên một kim loại, câu trả lời khoa học ngắn gọn là: liên kết kim loại cộng với cấu trúc tinh thể đều đặn tạo nên những tính chất quen thuộc mà chúng ta nhận ra.
Những mô hình nguyên tử này không chỉ kiểm soát độ bóng và độ bền. Chúng còn góp phần xác định những nguyên tố nào được xếp vào nhóm kim loại, và câu hỏi này dẫn thẳng tới bảng tuần hoàn cũng như vị trí tồn tại của kim loại có thể khai thác trong tự nhiên.
Vị trí của kim loại trên bảng tuần hoàn và trong tự nhiên
Cấu trúc kim loại giải thích hành vi của chúng, nhưng hóa học cũng phân loại kim loại dựa trên vị trí trong bảng tuần hoàn. Nếu bạn thắc mắc kim loại nằm ở đâu trên bảng tuần hoàn, câu trả lời ngắn gọn là phần lớn kim loại nằm ở phía bên trái và kéo dài qua vùng trung tâm của bảng. Các bảng tuần hoàn đặt các kim loại ở phía dưới và bên trái dải chéo của các bán kim loại, trong khi nhiều cột ở giữa là các nguyên tố chuyển tiếp, vốn cũng là kim loại.
Vị trí của kim loại trên bảng tuần hoàn
Bố cục này giúp trả lời đồng thời một số truy vấn phổ biến, bao gồm: kim loại nằm ở đâu trên bảng tuần hoàn, kim loại được đặt ở vị trí nào trên bảng tuần hoàn, và kim loại trong bảng tuần hoàn được tìm thấy ở đâu. Nói một cách đơn giản, hãy nhìn về phía bên trái để tìm các nhóm như kim loại kiềm và kim loại kiềm thổ, và quan sát phần trung tâm để tìm các kim loại chuyển tiếp như sắt, đồng và niken. Các phi kim tập trung ở góc trên bên phải, được phân tách khỏi kim loại bởi đường ranh giới dạng răng cưa quen thuộc.
Kim loại có nguồn gốc từ đâu trong tự nhiên
Một câu hỏi khác đặt ra là kim loại có nguồn gốc từ đâu. Trong tự nhiên, kim loại có thể sử dụng được thường bắt nguồn từ các mỏ quặng trong vỏ Trái Đất, chứ không phải từ những tấm, thanh hoặc bộ phận đã được chế tạo sẵn. Quặng là một mỏ tự nhiên chứa các khoáng chất có giá trị, và những khoáng chất này có thể chứa kim loại. Như Eagle Alloys lưu ý, kim loại thường được khai thác từ quặng, sau đó được chiết xuất và tinh luyện.
- Sắt thường được lấy từ quặng sắt.
- Nhôm thường được tìm thấy trong bôxít.
- Đồng được thu nhận từ các loại quặng đồng.
Tại sao quặng không giống với kim loại thành phẩm
Sự phân biệt này rất quan trọng. Một nguyên tố kim loại, chẳng hạn như nhôm hoặc sắt, là một nhóm trên bảng tuần hoàn . Quặng là một loại đá tự nhiên hoặc mỏ tự nhiên chứa các khoáng chất có chứa kim loại đó dưới dạng hợp chất hóa học. Vì vậy, khi ai đó hỏi kim loại bắt nguồn từ đâu, câu trả lời thực tiễn là từ quặng, trong khi câu trả lời về mặt hóa học lại hướng đến chính các nguyên tố kim loại. Chính sự trùng lặp về cách diễn đạt này dẫn đến việc nhiều người nhầm lẫn giữa kim loại nguyên chất, hợp kim, quặng, khoáng chất và hợp chất.

So sánh kim loại nguyên chất, hợp kim, quặng và hợp chất
Vị trí trên bảng tuần hoàn cho bạn biết nguyên tố đó là gì. Tuy nhiên, trong ngôn ngữ hàng ngày, người ta thường nói về vật liệu thay vì hóa học. Đó là lúc mọi người bắt đầu nhầm lẫn giữa một nguyên tố kim loại, một loại đá lấy từ lòng đất và một vật liệu kim loại đã qua chế tạo.
Kim loại nguyên chất so với hợp kim
Kim loại nguyên chất là một nguyên tố duy nhất được sử dụng dưới dạng vật liệu. Đồng, vàng và nhôm là những ví dụ điển hình. Về mặt hóa học, mỗi chất này là một yếu tố kim loại nguyên tố
Một hợp kim Kim loại hợp kim thì khác. Đây là một vật liệu dựa trên kim loại được tạo ra bằng cách kết hợp một kim loại nền với các nguyên tố khác nhằm thay đổi tính năng. Như Xometry giải thích, hợp kim thường bao gồm một kim loại nền cùng các thành phần kim loại hoặc phi kim được thêm vào. Vì vậy, thép, đồng thau và đồng đỏ không phải là kim loại nguyên chất, dù chúng rõ ràng thuộc loại kim loại trong sử dụng hàng ngày.
So sánh quặng, khoáng vật và hợp chất kim loại
| Danh mục | Định nghĩa | Thành phần cấu tạo | Có phải là nguyên tố trên bảng tuần hoàn? | Ví dụ quen thuộc |
|---|---|---|---|---|
| Kim loại nguyên chất | Một vật liệu gồm một nguyên tố duy nhất | Chỉ có một loại nguyên tử kim loại | Có | Đồng |
| HỢP KIM | Một vật liệu kim loại được thiết kế bằng cách trộn các nguyên tố | Một kim loại cơ sở cộng thêm các kim loại hoặc phi kim khác | No | Thép |
| Khoáng sản | Một chất kết tinh tự nhiên | Thành phần hóa học cụ thể và cấu trúc tinh thể | No | Hematit |
| Quặng | Một mỏ đá hoặc khoáng sản có giá trị khai thác để lấy kim loại | Một tập hợp giàu khoáng chất hoặc nguyên tố hữu ích đến mức đủ để khai thác mỏ | No | Bauxite |
| Hợp chất kim loại | Một chất có các nguyên tố liên kết với nhau bằng liên kết hóa học | Các nguyên tử kim loại liên kết với các nguyên tố khác | No | Oxit nhôm |
IBRAM tách riêng khoáng chất, đá, quặng và kim loại theo đúng cách này. Trung tâm Học tập Khoa học cũng lưu ý rằng phần lớn kim loại trong tự nhiên tồn tại dưới dạng hợp chất, chẳng hạn như oxit hoặc sunfua, và các hợp kim được sử dụng phổ biến hơn kim loại nguyên chất.
Cách phân biệt nguyên tố kim loại với vật liệu kim loại
Dưới đây là bài kiểm tra nhanh. Nếu nó có một ô trên bảng tuần hoàn, thì đó là một nguyên tố. Nếu nó là một vật liệu thực tế được chế tạo để sử dụng, thì nó có thể ở dạng nguyên chất hoặc là một hợp kim. Nếu nó được khai thác từ lòng đất, thì thường là quặng hoặc khoáng chất. Nếu kim loại liên kết hóa học với một chất khác, thì đó là một hợp chất.
Mọi người nhầm lẫn các thuật ngữ này vì một từ duy nhất — 'kim loại' — được sử dụng cả trong khoa học lẫn mua sắm. Cùng một người có thể gọi sắt là một nguyên tố, thép là một kim loại và bôxít là nguồn cung cấp kim loại trong cùng một cuộc trò chuyện. Cả ba khái niệm này đều có liên hệ với nhau, nhưng chúng không thuộc cùng một phạm trù. Sự khác biệt này còn quan trọng hơn nữa khi xem xét các tên gọi quen thuộc như sắt, thép, thép không gỉ, nhôm, đồng thau và đồng thanh, bởi mỗi tên gọi trả lời câu hỏi theo một cách hơi khác nhau.
Thép, nhôm, đồng thau và đồng thanh được cấu tạo từ những gì
Các tên gọi như sắt, thép, đồng và nhôm nghe có vẻ đơn giản, nhưng chúng không phải lúc nào cũng mô tả cùng một loại vật liệu. Một số là các nguyên tố tinh khiết; số khác là các hợp kim được tạo thành bằng cách pha trộn một kim loại nền với các nguyên tố khác. Đây là những ví dụ điển hình về các chất kim loại mà đa số người dân thường nghĩ đến khi đặt câu hỏi 'kim loại được cấu tạo từ những gì' trong đời sống hằng ngày.
Đó cũng là lý do vì sao các vật liệu phổ biến trong cửa hàng có thể trông tương tự nhau nhưng lại có tính chất vận hành rất khác biệt. Một sợi dây đồng, một bồn rửa inox và một phụ kiện đồng thau đều là các sản phẩm kim loại, thế nhưng thành phần cấu tạo của chúng khiến mỗi loại đảm nhiệm một vai trò khác nhau.
Các kim loại phổ biến và thành phần cấu tạo của chúng
| Vật liệu | Thành phần cấu tạo | Kim loại nguyên chất hoặc hợp kim | Cách thành phần ảnh hưởng đến các tính chất quen thuộc | Sử dụng phổ biến |
|---|---|---|---|---|
| Sắt | Chủ yếu gồm các nguyên tử sắt | Nguyên tố kim loại nguyên chất | Đóng vai trò kim loại nền cho nhiều vật liệu ferro (có chứa sắt). Khi thêm các nguyên tố khác, tính chất của nó thay đổi đáng kể. | Vật liệu nền để sản xuất thép, các thành phần từ tính |
| Thép | Sắt kết hợp với carbon, thường kèm theo các nguyên tố bổ sung như mangan, crôm, niken hoặc molypden | HỢP KIM | Carbon làm tăng độ bền của sắt, trong khi các nguyên tố bổ sung khác có thể cải thiện độ cứng, độ dai, khả năng hàn hoặc khả năng chống ăn mòn. | Dầm, bu lông và ốc vít, dụng cụ, phương tiện giao thông, bộ phận máy móc |
| Thép không gỉ | Sắt chứa crôm và thường cả niken, đôi khi có molipden | HỢP KIM | Crôm giúp tạo ra bề mặt chống ăn mòn mà mọi người thường liên hệ với các vật liệu không gỉ. | Bồn rửa, đồ dùng ăn uống, thiết bị chế biến thực phẩm, bộ phận y tế và hàng hải |
| Nhôm công nghiệp | Nguyên tử nhôm, mặc dù nhiều loại thương mại phổ biến được hợp kim hóa với magiê, silic, đồng, kẽm hoặc mangan | Nguyên tố kim loại tinh khiết trong hóa học, thường được hợp kim hóa trong thực tiễn | Khối lượng riêng thấp và khả năng chống ăn mòn tự nhiên khiến nó hữu ích trong các ứng dụng yêu cầu giảm trọng lượng. | Khung, tấm vỏ, lon đựng, bộ phận vận tải |
| Đồng | Chủ yếu gồm các nguyên tử đồng | Nguyên tố kim loại nguyên chất | Độ dẫn điện và dẫn nhiệt cao làm cho đồng trở nên quý giá, nhưng đồng tương đối mềm. | Dây điện, đầu nối, đường ống dẫn, bộ phận truyền nhiệt |
| Đồng thau | Đồng cộng với kẽm | HỢP KIM | So với đồng nguyên chất, đồng thau thường dễ gia công hơn và vẫn có khả năng chống ăn mòn khá tốt. | Các chi tiết lắp ghép, van, phụ kiện cơ khí, bộ phận trang trí |
| Đồng | Thường là đồng cộng với thiếc | HỢP KIM | Đồng thanh được đánh giá cao nhờ khả năng chịu mài mòn và hiệu suất ma sát thấp so với đồng mềm hơn. | Ổ bi, bạc lót, tấm chịu mài mòn, các vật thể được đúc |
Protolabs mô tả thép là một hợp kim sắt–cacbon, thường chứa từ 0,05% đến 2% cacbon theo trọng lượng, đồng thời lưu ý rằng thép không gỉ chứa ít nhất 10,5% crôm. MW Alloys phân loại đồng thau là hợp kim đồng–kẽm và đồng thanh là hợp kim đồng–thiếc, trong khi Các Thủ Thuật Thiết Kế Tự Động Hóa nhấn mạnh tính dẫn điện vượt trội của đồng và tính hữu dụng của đồng thanh trong các ứng dụng chịu mài mòn.
Thép được cấu tạo từ những nguyên tố nào so với nhôm và đồng
Nếu bạn đang thắc mắc thép được làm từ gì, câu trả lời ngắn gọn là sắt cộng thêm một lượng carbon được kiểm soát. Vậy kim loại nào có trong thép? Sắt là kim loại nền. Carbon có thể chỉ chiếm một phần nhỏ trong tổng thành phần, nhưng lại có ảnh hưởng lớn đến độ bền và độ cứng. Đó là lý do vì sao khi người ta hỏi thép được tạo thành từ những gì, thực chất họ đang tìm hiểu về công thức pha chế, chứ không chỉ đơn thuần là nguyên tố chính.
Một cách diễn đạt dễ hiểu: thành phần của thép thường bắt đầu từ sắt và carbon, sau đó được mở rộng khi kỹ sư cần đạt được các tính chất khác nhau. Mangan, niken, crôm và molypden là những nguyên tố thường được bổ sung vào nhiều loại thép. Nhôm và đồng cũng trả lời cùng một câu hỏi nhưng theo một cách khác. Nhôm là một nguyên tố hóa học, tuy nhiên nhiều bộ phận nhôm trong thực tế lại là hợp kim. Đồng cũng là một nguyên tố hóa học và vẫn giữ vai trò quan trọng khi độ dẫn điện là yếu tố ưu tiên hơn độ bền cao.
Cách thành phần hợp kim thay đổi tính chất và ứng dụng
Những thay đổi nhỏ trong thành phần có thể tạo ra các vật liệu rất khác nhau. Thêm carbon vào sắt, bạn sẽ thu được thép. Thêm đủ crôm vào loại thép đó, bạn sẽ có thép không gỉ. Trộn đồng với kẽm, bạn sẽ được đồng thau. Trộn đồng với thiếc, bạn sẽ được đồng thanh. Đó là lý do vì sao các loại kim loại khác nhau có thể phục vụ những mục đích hoàn toàn khác nhau, ngay cả khi chúng đều trông đơn giản như kim loại thông thường trước mắt.
- Hàm lượng carbon cao hơn trong thép thường làm tăng độ cứng và độ bền, nhưng có thể khiến việc gia công và hàn trở nên khó khăn hơn.
- Crom trong thép không gỉ cải thiện khả năng chống ăn mòn bằng cách hỗ trợ hình thành một lớp bề mặt bảo vệ.
- Kẽm trong đồng thau nâng cao khả năng gia công cơ khí, nhờ đó đồng thau được sử dụng phổ biến trong các chi tiết lắp ghép và phụ kiện.
- Thiếc trong đồng thanh cải thiện tính chịu mài mòn, điều này giải thích cho việc đồng thanh thường được dùng trong các ổ trượt và bạc lót.
Tên trên một sản phẩm hoàn chỉnh cho bạn biết danh mục vật liệu, nhưng không phản ánh toàn bộ hành trình đằng sau nó. Thép, nhôm và đồng không bắt đầu dưới dạng dầm, tấm hay dây. Trước khi trở thành nguyên vật liệu hữu dụng, chúng phải được khai thác, tinh luyện và đôi khi được trộn theo tỷ lệ nhất định để tạo thành dạng thức mà người ta thường nhận ra.
Quy trình sản xuất kim loại từ quặng thành vật liệu hoàn chỉnh
Một dầm thép hoặc cuộn dây đồng trông đơn giản khi đã đến kho hàng hoặc nhà máy. Tuy nhiên, hành trình dẫn đến sản phẩm đó lại hết sức phức tạp. Trong lòng đất, kim loại có ích thường bị khóa bên trong quặng dưới dạng một hợp chất. Sau đó, nó được chiết xuất thành kim loại nguyên chất. Và sau đó nữa, nó có thể được pha trộn thành hợp kim rồi gia công thành sản phẩm sử dụng được.
Người ta thường tìm kiếm các cụm từ như 'kim loại được sản xuất như thế nào', 'kim loại được làm ra ra sao' hoặc 'chúng ta sản xuất kim loại bằng cách nào'. Câu trả lời thực tế là một chuỗi các bước, và mỗi bước đều làm thay đổi thành phần cấu tạo của vật liệu.
Quy trình sản xuất kim loại từ quặng
- Phát hiện quặng: Các nhà địa chất xác định các cấu tạo đá chứa khoáng chất có giá trị. Quặng là loại đá chứa các khoáng chất quan trọng có kim loại hữu ích bên trong.
- Khai thác mỏ: Quặng được khai thác từ lòng đất và đưa đi xử lý.
- Sàng lọc, nghiền và xay nhỏ: Đá được đập vỡ thành những mảnh nhỏ hơn để phần có giá trị có thể được tách ra hiệu quả hơn. Metal Supermarkets mô tả đây là những bước chuẩn bị ban đầu trong quá trình khai thác.
- : Vật liệu thải (gọi là đá kèm), được giảm thiểu nhằm làm tăng hàm lượng vật liệu mang kim loại trong quặng.
- Nung nóng hoặc nung phân hủy: Nhiều loại quặng cần được nung nóng trước khi kim loại có thể được giải phóng. CK-12 giải thích rằng quặng sunfua thường được nung trong không khí, trong khi quặng cacbonat được nung phân hủy trong điều kiện thiếu hoặc không có không khí, thường để tạo thành các oxit kim loại.
- Chiết xuất và luyện kim: Ở giai đoạn chiết xuất ở nhiệt độ cao, hợp chất kim loại được chuyển thành kim loại nguyên chất. Tùy thuộc vào tính phản ứng, quá trình này có thể diễn ra thông qua phản ứng khử bằng carbon hoặc hydro, phản ứng thế bởi một kim loại phản ứng mạnh hơn, hoặc điện phân muối nóng chảy đối với các kim loại có tính phản ứng rất cao.
- Tinh luyện: Kim loại thu được ban đầu thường chưa tinh khiết. Quá trình tinh luyện loại bỏ thêm các tạp chất không mong muốn và nâng cao độ tinh khiết.
- Hợp kim hóa và tạo hình: Nếu cần thiết, các nguyên tố khác sẽ được bổ sung, và kim loại được tạo hình thành tấm, thanh, dây hoặc các chi tiết hoàn chỉnh.
Từ khai thác và luyện kim đến tinh luyện
Cách kim loại được sản xuất là điều quan trọng vì đặc điểm của vật liệu thay đổi dọc theo quy trình. Trước khi khai thác, vật liệu chủ yếu là hợp chất kim loại trộn lẫn với đá và các tạp chất. Sau khi khử hoặc điện phân, vật liệu trở thành kim loại, nhưng chưa hoàn toàn sạch. Quá trình tinh luyện đưa nó gần hơn tới dạng kim loại nguyên tố tinh khiết. Trong tinh luyện điện phân, CK-12 ghi nhận rằng kim loại di chuyển từ cực dương (anode) không tinh khiết và được lắng đọng lên cực âm (cathode) tinh khiết.
Cách kim loại tinh khiết trở thành vật liệu hợp kim
Kim loại nguyên chất không phải lúc nào cũng là mục tiêu cuối cùng. Sắt có thể được hợp kim hóa với carbon để tạo thành thép. Đồng có thể được trộn với kẽm để tạo thành đồng thau. Nhôm cũng được sử dụng rộng rãi dưới dạng các hợp kim. Vì vậy, khi ai đó hỏi kim loại được sản xuất như thế nào, họ thực sự có thể đang muốn biết về kim loại trong quặng, kim loại sau khi chiết xuất, hoặc kim loại sau khi hợp kim hóa thành một vật liệu thực tiễn.
Chính sự thay đổi nghĩa này là lý do vì sao những phát biểu thường ngày về thép, thép không gỉ, carbon và gỉ sắt thường đòi hỏi phải xem xét kỹ lưỡng hơn.
Thép là kim loại hay là nguyên tố?
Đây chính là điểm khiến khái niệm kim loại trở nên khó hiểu đối với nhiều người mới bắt đầu. Trong lời nói hằng ngày, người ta thường nhầm lẫn giữa các nguyên tố, hợp kim và quá trình ăn mòn như thể chúng là một thứ. Đó là lý do vì sao mọi người đặt câu hỏi: thép có phải là kim loại không, thép có phải là nguyên tố không, hoặc thậm chí là phiên bản đảo ngược: kim loại có phải là thép không.
Thép là kim loại hay là nguyên tố
Thép là một vật liệu kim loại, nhưng không phải là một nguyên tố trong bảng tuần hoàn. Thép là một hợp kim chủ yếu gồm sắt và carbon.
Cách đơn giản nhất để làm rõ vấn đề này là tách biệt khái niệm hóa học với vật liệu. Sắt là kim loại nguyên tố nằm ở nền tảng của thép. Thép là một vật liệu được sản xuất từ sắt đó. Các mô tả tiêu chuẩn về thành phần thép nêu rõ rằng thép chủ yếu gồm sắt và carbon, thường chiếm khoảng 0,02% đến 2,14% theo khối lượng. Vì vậy, câu trả lời cho câu hỏi "thép có phải là kim loại không?" là có. Còn câu trả lời cho câu hỏi "thép có phải là nguyên tố không?" là không.
Lập luận tương tự cũng áp dụng để trả lời câu hỏi "thép không gỉ có phải là kim loại không?" — Câu trả lời là có. Thép không gỉ vẫn là thép, chỉ khác ở công thức hợp kim. Các nguồn tài liệu về thép không gỉ và các loại thép ghi nhận rằng các mác thép không gỉ thường chứa hơn 10,5% crôm, giúp cải thiện khả năng chống ăn mòn.
Tại sao carbon lại làm thay đổi tính chất của kim loại mà bản thân nó lại không phải là kim loại
Nếu bạn đã tìm kiếm thông tin về carbon là kim loại hay phi kim, thì câu trả lời ngắn gọn là: carbon là phi kim. Dẫu vậy, carbon có thể làm thay đổi mạnh mẽ cách sắt hoạt động khi cả hai được kết hợp với nhau trong thép. Trong thép cacbon, hàm lượng cacbon cao hơn sẽ làm tăng độ cứng nhưng đồng thời làm giảm độ dẻo dai, như minh họa trong bảng so sánh thép cacbon. Đây là một lời nhắc nhở hữu ích rằng một nguyên tố hợp kim không nhất thiết phải là kim loại mới có thể làm thay đổi tính chất của kim loại.
Những nhận định phổ biến về kim loại cần được điều chỉnh
- Hiểu lầm: Thép là một kim loại nguyên chất riêng biệt. Thông tin: Thép là một hợp kim của sắt và cacbon, thường chứa thêm các nguyên tố khác.
- Hiểu lầm: Thép không gỉ thực tế không phải là kim loại. Thông tin: Thép không gỉ vẫn là một hợp kim kim loại.
- Hiểu lầm: Sắt và thép là một và cùng một thứ. Thông tin: Sắt là nguyên tố nền, còn thép là một vật liệu được chế tạo từ sắt.
- Hiểu lầm: Gỉ sét giống hệt kim loại. Thông tin: Gỉ sét mô tả tình trạng ăn mòn trên bề mặt, chứ không phải là danh mục phân loại kim loại.
- Hiểu lầm: Kim loại được cấu tạo từ các nguyên tử, vì vậy chúng không xuất phát từ quặng. Thông tin: Cả hai quan điểm đều đúng. Một quan điểm mô tả kim loại ở cấp độ nguyên tử, còn quan điểm kia mô tả nguồn gốc của kim loại có thể sử dụng được trước khi tiến hành khai thác và tinh luyện.
Những sai sót nhỏ trong cách diễn đạt có thể dẫn đến những hiểu lầm lớn về vật liệu, đặc biệt là khi thành phần bắt đầu ảnh hưởng đến độ bền, khả năng chống ăn mòn, khả năng gia công và cách thức chế tạo các chi tiết thực tế.

Thành phần kim loại định hướng các lựa chọn sản xuất thực tế
Trong nhà máy, yếu tố hóa học nhanh chóng thoát khỏi tính trừu tượng. Ngay khoảnh khắc một chi tiết cần được cắt, uốn, dập hoặc hoàn thiện bề mặt, trọng tâm câu hỏi sẽ chuyển từ việc kim loại được cấu tạo bởi những gì sang việc thành phần đó sẽ phản ứng ra sao trong quá trình sản xuất cũng như trong điều kiện vận hành thực tế. Các loại kim loại khác nhau có thể trông tương tự nhau trên giấy, nhưng lại thể hiện hiệu suất rất khác biệt khi chịu tác động của nhiệt, lực, độ ẩm và dung sai chặt chẽ.
Thành phần kim loại định hướng hiệu suất của chi tiết
Hướng dẫn lựa chọn vật liệu từ Sinoway cho thấy lý do vì sao điều này quan trọng: độ cứng, độ bền va đập, độ dẻo, độ dẫn nhiệt và khả năng chống ăn mòn đều ảnh hưởng đến hành vi gia công, mài mòn dụng cụ, độ nhẵn bề mặt và chất lượng cuối cùng. Nói cách khác, các đặc tính của kim loại không chỉ là những dữ kiện trong phòng thí nghiệm; chúng trực tiếp ảnh hưởng đến chi phí, tốc độ, độ bền và độ ổn định.
- Độ bền và độ cứng: các vật liệu cứng hơn có thể chịu được tải trọng yêu cầu cao, nhưng thường làm tăng mức độ mài mòn dụng cụ và làm chậm quá trình cắt.
- Khả năng chống ăn mòn: thép không gỉ và nhôm thường được ưa chuộng trong các ứng dụng liên quan đến độ ẩm hoặc môi trường khắc nghiệt.
- Khả năng gia công: nhôm được sử dụng rộng rãi khi yêu cầu tốc độ cắt nhanh và hình học phức tạp.
- Khả năng định hình: độ dẻo hỗ trợ quá trình tạo hình, tuy nhiên các vật liệu quá dẻo có thể gây khó khăn hơn trong việc kiểm soát kích thước.
- Tính dẫn điện: đồng vẫn giữ giá trị cao trong các ứng dụng đòi hỏi truyền nhiệt hoặc dẫn điện.
- Chất lượng bề mặt: thành phần hóa học ảnh hưởng đến độ hoàn thiện và độ chính xác đạt được của chi tiết.
Lựa chọn phương pháp gia công kim loại cho các ứng dụng thực tế
Hướng dẫn lựa chọn vật liệu của LS Manufacturing tập trung vào độ bền, trọng lượng, điều kiện môi trường, khả năng gia công cơ khí và chi phí. Đây là cách thực tiễn để trả lời câu hỏi 'kim loại được sử dụng để làm gì?'. Một giá đỡ nhẹ có thể ưu tiên nhôm. Một bộ phận tiếp xúc với môi trường ăn mòn có thể thiên về thép không gỉ. Một bộ phận dẫn điện có thể yêu cầu đồng. Các tính chất chính của kim loại chỉ trở nên hữu ích khi được kết hợp phù hợp với yêu cầu thực tế của công việc.
Khi nào nên hợp tác với đối tác sản xuất
Khi các mục tiêu hiệu suất, dung sai và khối lượng sản xuất đều quan trọng cùng lúc, việc lựa chọn vật liệu sẽ trở thành một quyết định quy trình nhiều hơn là một quyết định dựa trên thành phần hóa học. Đối với các nhà sản xuất ô tô và nhà cung cấp cấp 1, Shaoyi là một ví dụ minh họa rõ ràng cho bước tiếp theo này, cung cấp dịch vụ dập chính xác cao, gia công CNC, chế tạo mẫu nhanh, xử lý bề mặt tùy chỉnh và sản xuất ô tô khối lượng lớn dưới hệ thống đảm bảo chất lượng IATF 16949. Những độc giả cần hỗ trợ triển khai có thể xem xét các dịch vụ của Shaoyi tại dịch Vụ . Đó là lúc việc hiểu rõ thành phần cấu tạo của kim loại cuối cùng được chuyển hóa thành những chi tiết đáng tin cậy trên dây chuyền sản xuất.
Các câu hỏi thường gặp về thành phần cấu tạo của kim loại
1. Kim loại được cấu tạo từ những gì, nói một cách đơn giản?
Nói một cách đơn giản, kim loại được cấu tạo từ các nguyên tử kim loại sắp xếp theo một cấu trúc rắn. Trong tự nhiên, những nguyên tử này thường bị bao bọc trong quặng hoặc khoáng vật, do đó kim loại thường phải được chiết xuất trước tiên. Trong đời sống hằng ngày, vật liệu cuối cùng có thể là một kim loại nguyên chất như đồng hoặc một hợp kim như thép.
2. Kim loại có nguồn gốc từ đâu trong tự nhiên?
Hầu hết kim loại có thể sử dụng được bắt nguồn từ các mỏ quặng nằm trong lòng đất. Việc khai thác và chế biến sẽ tách vật liệu chứa kim loại có giá trị ra khỏi đá; sau đó, quá trình chiết xuất và tinh luyện biến chúng thành kim loại có thể sử dụng được. Một số ít kim loại có thể tồn tại ở dạng kim loại tự nhiên, nhưng đa số kim loại công nghiệp đến với chúng ta thông qua quy trình chuyển đổi từ quặng thành kim loại.
3. Sự khác biệt giữa kim loại nguyên chất, hợp kim và quặng là gì?
Một kim loại nguyên chất là một nguyên tố hóa học được sử dụng làm vật liệu, ví dụ như nhôm hoặc đồng. Một hợp kim là hỗn hợp dựa trên kim loại được chế tạo nhằm cải thiện các tính chất, ví dụ như thép, đồng thau hoặc đồng đỏ. Một quặng hoàn toàn không phải là kim loại thành phẩm, mà là vật liệu nguồn tự nhiên chứa các hợp chất hoặc khoáng chất từ đó kim loại có thể được chiết xuất.
4. Thép được cấu tạo từ những thành phần nào, và thép có phải là một nguyên tố hay không?
Thép chủ yếu được sản xuất từ sắt và carbon; nhiều loại thép còn chứa thêm các nguyên tố như crôm, niken hoặc mangan. Những thành phần bổ sung này làm thay đổi hiệu suất của vật liệu, bao gồm độ cứng, độ dai và khả năng chống ăn mòn. Thép rõ ràng là một vật liệu kim loại, nhưng không phải là một nguyên tố trong bảng tuần hoàn vì thép là một hợp kim chứ không phải một nguyên tố duy nhất.
5. Tại sao thành phần kim loại lại quan trọng trong sản xuất?
Thành phần ảnh hưởng đến cách một kim loại được cắt, uốn, dập, hàn và xử lý bề mặt, cũng như khả năng chống mài mòn hoặc ăn mòn. Điều này có nghĩa là việc lựa chọn vật liệu tác động đến cả hiệu suất của chi tiết và hiệu quả sản xuất. Đối với các chương trình ô tô cần hỗ trợ chuyển kiến thức về vật liệu thành các linh kiện thực tế, một đối tác như Shaoyi có thể cung cấp dịch vụ dập, gia công CNC, chế tạo mẫu, xử lý bề mặt và sản xuất hàng loạt theo hệ thống chất lượng IATF 16949.
 Sản xuất với số lượng nhỏ, tiêu chuẩn cao. Dịch vụ tạo nguyên mẫu nhanh của chúng tôi giúp việc kiểm chứng trở nên nhanh chóng và dễ dàng hơn —
Sản xuất với số lượng nhỏ, tiêu chuẩn cao. Dịch vụ tạo nguyên mẫu nhanh của chúng tôi giúp việc kiểm chứng trở nên nhanh chóng và dễ dàng hơn —
